Sam Brusco, Associate Editor03.08.22
Dental artificial intelligence (AI) solutions firm Pearl has earned U.S. Food and Drug Administration (FDA) clearance for its AI-powered, real-time pathology detection tool Second Opinion. The solution helps identify a broad range of common dental conditions in patient x-rays.
According to the company, Second Opinion is now the only AI-driven computer-aided clinical radiology tool for dentists throughout North America, Europe, Australia, and other areas of the globe.
“This clearance is a major milestone not only for our team and for the many dentists, advisors and partners who have contributed to Second Opinion’s development, but also for dentistry itself,” Pearl CEO and founder Ophir Tanz told the press. “AI is a paradigm-shifting technology that will add value across the entire healthcare continuum. Because x-rays are a regular part of every dental patient’s experience, the first place most people will encounter the power of medical AI technology will be in their dentist’s chair. Second Opinion’s FDA clearance has made that possible.”
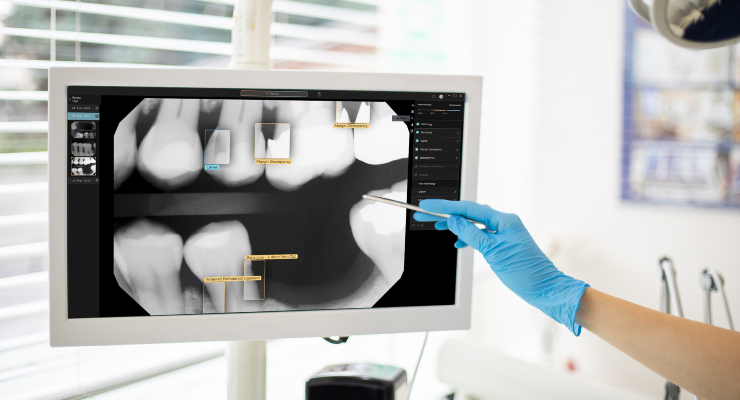
The system applies computer vision to identify and highlight dental caries, discrepancies at the margin of existing restorations, calculus, periapical radiolucency, crowns, fillings, root canals, bridges, and implants. After x-rays are captured, Second Opinion displays images and any detected conditions on monitors in the operatory.
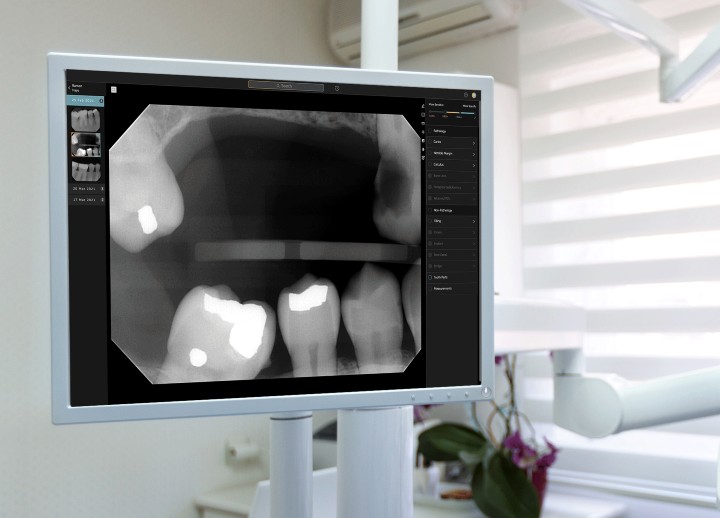
Second Opinion features a clear, high-contrast user interface for optimal X-ray viewability.
“The benefit that Pearl’s AI brings to patient communication in the dental operatory – and the trust that follows—cannot be overstated, and it is in that area that Second Opinion’s impact will be most immediately felt,” said Professor Dr. Markus Blatz, DMD, a leading authority in restorative dentistry and digital innovation. “Its long-term impact on the field of clinical dentistry may be more important, however. We endeavor to establish a standard of care through instruction at dental school, but at some point our instruction stops. Second Opinion will help us maintain that standard and, over time, establish a more consistently rewarding patient experience.”
Clinical studies supporting the FDA clearance featured radiographic datasets of over 2,000 images and reader group of 86 exert dentists and dental radiologists. Study participants reading x-rays with Second Opinion’s assistance accurately spotted 36 percent more lesions than those without AI assistance.
“Second Opinion now joins a family of FDA-cleared CADe medical systems already in use for radiologically-driven tasks such as lung nodule detection and mammography interpretation,” Pearl’s chief technology officer and co-founder Cambron Carter said. “State of the art algorithms that currently assist in the detection of cancerous lesions can now be applied to detect many more frequently occurring dental diseases. The standard of care in dentistry is about to level up.”
According to the company, Second Opinion is now the only AI-driven computer-aided clinical radiology tool for dentists throughout North America, Europe, Australia, and other areas of the globe.
“This clearance is a major milestone not only for our team and for the many dentists, advisors and partners who have contributed to Second Opinion’s development, but also for dentistry itself,” Pearl CEO and founder Ophir Tanz told the press. “AI is a paradigm-shifting technology that will add value across the entire healthcare continuum. Because x-rays are a regular part of every dental patient’s experience, the first place most people will encounter the power of medical AI technology will be in their dentist’s chair. Second Opinion’s FDA clearance has made that possible.”
The system applies computer vision to identify and highlight dental caries, discrepancies at the margin of existing restorations, calculus, periapical radiolucency, crowns, fillings, root canals, bridges, and implants. After x-rays are captured, Second Opinion displays images and any detected conditions on monitors in the operatory.

Second Opinion features a clear, high-contrast user interface for optimal X-ray viewability.
“The benefit that Pearl’s AI brings to patient communication in the dental operatory – and the trust that follows—cannot be overstated, and it is in that area that Second Opinion’s impact will be most immediately felt,” said Professor Dr. Markus Blatz, DMD, a leading authority in restorative dentistry and digital innovation. “Its long-term impact on the field of clinical dentistry may be more important, however. We endeavor to establish a standard of care through instruction at dental school, but at some point our instruction stops. Second Opinion will help us maintain that standard and, over time, establish a more consistently rewarding patient experience.”
Clinical studies supporting the FDA clearance featured radiographic datasets of over 2,000 images and reader group of 86 exert dentists and dental radiologists. Study participants reading x-rays with Second Opinion’s assistance accurately spotted 36 percent more lesions than those without AI assistance.
“Second Opinion now joins a family of FDA-cleared CADe medical systems already in use for radiologically-driven tasks such as lung nodule detection and mammography interpretation,” Pearl’s chief technology officer and co-founder Cambron Carter said. “State of the art algorithms that currently assist in the detection of cancerous lesions can now be applied to detect many more frequently occurring dental diseases. The standard of care in dentistry is about to level up.”