Sam Brusco, Associate Editor02.18.22
Medtronic’s Freezor Xtra cardiac cryoablation catheters today gained expanded U.S. Food and Drug Administration (FDA) approval for pediatric atrioventricular nodal reentrant tachycardia (AVNRT). AVNRT causes very rapid heart rhythm and left untreated affects normal heart pumping, causing palpitations, lightheadedness, and syncope.
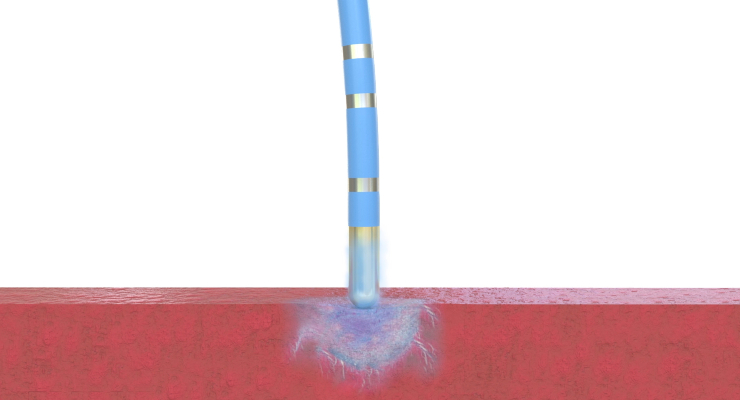
The flexible, single-use Freezor Xtra catheters freeze heart tissue to block unnecessary heart signals. Cryoablation can reduce risk of permanent AV black, a risk of AVNRT surgeries using RF ablations that results in heart electrical signal interruption and disrupts rhythm.
"There are very few devices approved to treat medically complex pediatric cardiology patients today," Bryan C. Cannon, M.D., professor of pediatrics and past president of the Pediatric & Congenital Electrophysiology Society (PACES) told the press. "With an FDA indication expansion, the Freezor and Freezor Xtra cardiac cryoablation catheters allow even the youngest of cardiology patients access to a safe, life-enhancing technology that will help advance cardiac care for AVNRT."
The approval was supported by the ICY-AVNRT trial and other pediatric studies, which determined 95 percent success with no reports of permanent pacemaker implant due to complete AV block.
The Freezor cryoablation catheter franchise was first introduced for U.S. adults with AVNRT in 2003. The next generation, Freezor Xtra, launched in 2016.
"We're proud of our work with PACES and FDA in this first-of-its-kind, multi-stakeholder initiative to address a critical patient population," said Rebecca Seidel, president of Medtronic’s Cardiac Ablation Solutions business. "The shared commitment to collaborate and grow this therapy's unique position to treat AVNRT patients demonstrates our confidence in the proven safety and efficacy of our cryoablation technology."
The flexible, single-use Freezor Xtra catheters freeze heart tissue to block unnecessary heart signals. Cryoablation can reduce risk of permanent AV black, a risk of AVNRT surgeries using RF ablations that results in heart electrical signal interruption and disrupts rhythm.
"There are very few devices approved to treat medically complex pediatric cardiology patients today," Bryan C. Cannon, M.D., professor of pediatrics and past president of the Pediatric & Congenital Electrophysiology Society (PACES) told the press. "With an FDA indication expansion, the Freezor and Freezor Xtra cardiac cryoablation catheters allow even the youngest of cardiology patients access to a safe, life-enhancing technology that will help advance cardiac care for AVNRT."
The approval was supported by the ICY-AVNRT trial and other pediatric studies, which determined 95 percent success with no reports of permanent pacemaker implant due to complete AV block.
The Freezor cryoablation catheter franchise was first introduced for U.S. adults with AVNRT in 2003. The next generation, Freezor Xtra, launched in 2016.
"We're proud of our work with PACES and FDA in this first-of-its-kind, multi-stakeholder initiative to address a critical patient population," said Rebecca Seidel, president of Medtronic’s Cardiac Ablation Solutions business. "The shared commitment to collaborate and grow this therapy's unique position to treat AVNRT patients demonstrates our confidence in the proven safety and efficacy of our cryoablation technology."