Sam Brusco, Associate Editor01.28.21
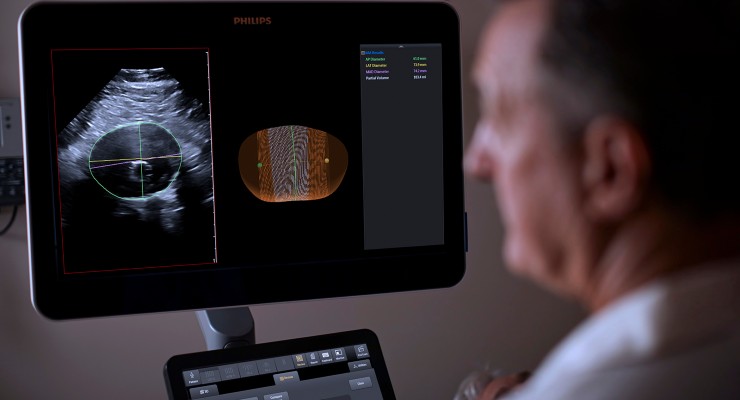
Philips has introduced the Philips Abdominal Aortic Aneurysm (AAA) Model. Based on 3D ultrasound, Philips AAA Model provides accurate diagnostic information without exposing patients to high radiation doses and nephrotoxic contrast agents.
AAAs are usually identified incidentally during abdominal imaging exams but sometimes go unnoticed until rupture. A ruptured AAA has an 80 percent mortality rate1. Philips AAA Model integrates software and the firm’s proprietary 3D ultrasound technologies into a single solution to help increase diagnostic confidence and improve patient experience. The software automatically segments and quantifies the size of the aneurysm sac for surveillance of known native and post-EVAR AAAs.

A recent clinical study showed 3D ultrasound examination for native AAA surveillance has excellent inter-operator reproducibility, superior to 2D ultrasound, supporting the broader use of 3D ultrasound in standard AAA surveillance programs2. 3D ultrasound has been shown to estimate the diameter and volume of an AAA with acceptable reproducibility and an improved agreement (over 2D ultrasound) with CT3. 3D ultrasound has also been proven to correlate significantly better to 3D CT than 2D ultrasound for assessing the residual sac’s maximum diameter post-EVAR, with clinically acceptable reproducibility4.
“Regular surveillance of abdominal aortic aneurysm patients is essential, but today’s standard of care has downsides,” Bich Le, senior VP and GM, Ultrasound at Philips. “Philips Abdominal Aortic Aneurysm (AAA) Model seamlessly integrates leading Philips technologies including Philips Premium Ultrasound System (EPIQ Elite), Philips Array Transducer (X6-1 xMATRIX) and innovative software into a single solution, to help improve the experience and outcomes for clinicians and patients alike.”

The Philips AAA Model is CE marked and is FDA cleared.
References
1 Age-specific incidence, risk factors, and outcome of acute abdominal aortic aneurysms in a defined population. Howard DP, Banerjee A, Fairhead JF, et al. Br J Surg. 2015;102(8):907–915. doi:10.1002/bjs.9838
2 Clinical validation of three-dimensional ultrasound for abdominal aortic aneurysm. Ghulam, Qasam M. et al., Journal of Vascular Surgery, Volume 71, Issue 1
3 Three-dimensional ultrasound evaluation of small asymptomatic abdominal aortic aneurysms. Bredahl, K. et al. European Journal of Vascular and Endovascular Surgery, Volume 49, Issue 3, 289 – 296
4 Three-dimensional ultrasound improves the accuracy of diameter measurement of the residual sac in EVAR patients. K. Bredahl, M. Taudorf, A. Long, L. Lönn, L. Rouet, R. Ardon, H. Sillesen, J.P. Eiberg. European Journal of Vascular and Endovascular Surgery, Volume 46, Issue 5, 2013
AAAs are usually identified incidentally during abdominal imaging exams but sometimes go unnoticed until rupture. A ruptured AAA has an 80 percent mortality rate1. Philips AAA Model integrates software and the firm’s proprietary 3D ultrasound technologies into a single solution to help increase diagnostic confidence and improve patient experience. The software automatically segments and quantifies the size of the aneurysm sac for surveillance of known native and post-EVAR AAAs.

A recent clinical study showed 3D ultrasound examination for native AAA surveillance has excellent inter-operator reproducibility, superior to 2D ultrasound, supporting the broader use of 3D ultrasound in standard AAA surveillance programs2. 3D ultrasound has been shown to estimate the diameter and volume of an AAA with acceptable reproducibility and an improved agreement (over 2D ultrasound) with CT3. 3D ultrasound has also been proven to correlate significantly better to 3D CT than 2D ultrasound for assessing the residual sac’s maximum diameter post-EVAR, with clinically acceptable reproducibility4.
“Regular surveillance of abdominal aortic aneurysm patients is essential, but today’s standard of care has downsides,” Bich Le, senior VP and GM, Ultrasound at Philips. “Philips Abdominal Aortic Aneurysm (AAA) Model seamlessly integrates leading Philips technologies including Philips Premium Ultrasound System (EPIQ Elite), Philips Array Transducer (X6-1 xMATRIX) and innovative software into a single solution, to help improve the experience and outcomes for clinicians and patients alike.”

The Philips AAA Model is CE marked and is FDA cleared.
References
1 Age-specific incidence, risk factors, and outcome of acute abdominal aortic aneurysms in a defined population. Howard DP, Banerjee A, Fairhead JF, et al. Br J Surg. 2015;102(8):907–915. doi:10.1002/bjs.9838
2 Clinical validation of three-dimensional ultrasound for abdominal aortic aneurysm. Ghulam, Qasam M. et al., Journal of Vascular Surgery, Volume 71, Issue 1
3 Three-dimensional ultrasound evaluation of small asymptomatic abdominal aortic aneurysms. Bredahl, K. et al. European Journal of Vascular and Endovascular Surgery, Volume 49, Issue 3, 289 – 296
4 Three-dimensional ultrasound improves the accuracy of diameter measurement of the residual sac in EVAR patients. K. Bredahl, M. Taudorf, A. Long, L. Lönn, L. Rouet, R. Ardon, H. Sillesen, J.P. Eiberg. European Journal of Vascular and Endovascular Surgery, Volume 46, Issue 5, 2013