FineHeart01.18.21
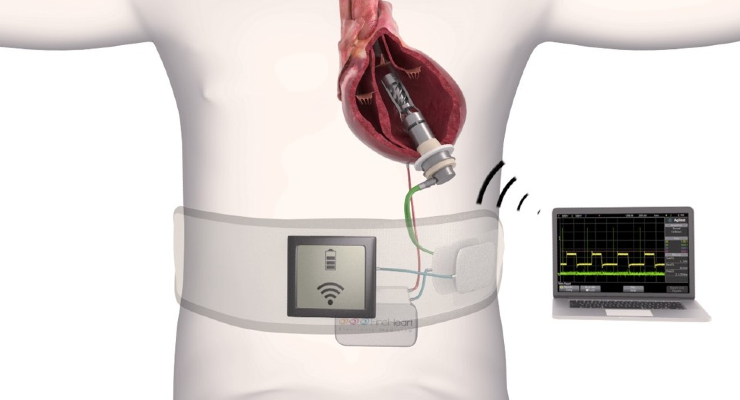
FineHeart, SA, a preclinical-stage medical device company developing an Implantable Cardiac Output Management System (ICOMS) to address the unmet need of patients suffering from severe heart failure, has completed a seven-day study of its Transcutaneous Energy Transfer (TET) System for recharging the ICOMS implantable battery.
"We are delighted to have succeeded in this major technological advancement proving the ability to power a Cardiac Assist device via an intelligent and minimally invasive TET system. This is a breakthrough in treating patients with severe heart failure who will be able to regain a quasi-normal quality of life without any device connected to their skin," said Arnaud Mascarell, CEO and founder, FineHeart. "We are very pleased with the success of this study, which is a world-first: to date, no TET system of this size supporting the pulsatility function has been realized. This study marks another crucial milestone for FineHeart and further validates the design freeze of our technology."
The trial confirms the TET’s ability to operate and provide power to the miniature heart pump in pulsatile mode and makes it one of the smallest TET system studies ever completed in this domain. The energy transfer system was safely tested as quickly implantable (15 minutes), having low energy consumption and avoiding tissue heat absorption. These benefits will ease recovery for already fragile patients. Furthermore, eliminating an external driveline removes the main cause of infection and complications for patients implanted with an LVAD and will reduce morbidity and mortality rates.
The ICOMS intracardiac location and its ability to synchronize with the heart's natural contractions make it exceptionally energy efficient. ICOMS is designed to support, rather than replace, especially not to go against the natural blood flow direction and fight against very high-pressure gradients, as the devices currently on the market do.
ICOMS 'feeds off' the intracardiac force generated with each contraction and relies on their strength to create additional and therapeutic blood flow. While respecting native cardiac flows, this support allows a significant reduction in its consumption, up to five times smaller than that observed in the Cardiac Assistance Devices (DAVG) currently on the market.
About the In-Vivo Study
The TET system was implanted for seven days, with five temperature sensors located throughout the internal coil pocket. The inner coil diameter is 8cm at 0.8 cm thick (same dimensions as an internal defibrillator, a device implanted daily in the G8 countries).
Heating was monitored for seven days with a consumption profile corresponding to a system operating at full speed, under extreme consumption conditions, well above nominal modes. The study showed that the average heating increase was around 1.5 +/- 0.2 °C, resulting in no tissue damage and no infection.
“The results of this study are remarkable and demonstrate that this small, mini-invasive TET system is functional and compatible with FDA guidelines and current regulations,” said professor Michael Acker, M.D., head of the Cardiac Surgery at the University of Pennsylvania Medical Center and FineHeart scientific advisory board member. “The ICOMS technology’s unique ability to replicate the volumetric displacement of the blood, beat by beat, by generating physiological cardiac flows addresses a significant unmet clinical need and could improve the quality of life for a huge population suffering from severe heart failure today and tomorrow.”
Heart failure (H.F.) is the second leading cause of death in the U.S. and Europe, a global pandemic affecting at least 26 million people worldwide and increasing prevalence. It is a degenerative disease leading to poor quality of life, frequent, costly hospitalizations, and early mortality.
"We are delighted to have succeeded in this major technological advancement proving the ability to power a Cardiac Assist device via an intelligent and minimally invasive TET system. This is a breakthrough in treating patients with severe heart failure who will be able to regain a quasi-normal quality of life without any device connected to their skin," said Arnaud Mascarell, CEO and founder, FineHeart. "We are very pleased with the success of this study, which is a world-first: to date, no TET system of this size supporting the pulsatility function has been realized. This study marks another crucial milestone for FineHeart and further validates the design freeze of our technology."
The trial confirms the TET’s ability to operate and provide power to the miniature heart pump in pulsatile mode and makes it one of the smallest TET system studies ever completed in this domain. The energy transfer system was safely tested as quickly implantable (15 minutes), having low energy consumption and avoiding tissue heat absorption. These benefits will ease recovery for already fragile patients. Furthermore, eliminating an external driveline removes the main cause of infection and complications for patients implanted with an LVAD and will reduce morbidity and mortality rates.
The ICOMS intracardiac location and its ability to synchronize with the heart's natural contractions make it exceptionally energy efficient. ICOMS is designed to support, rather than replace, especially not to go against the natural blood flow direction and fight against very high-pressure gradients, as the devices currently on the market do.
ICOMS 'feeds off' the intracardiac force generated with each contraction and relies on their strength to create additional and therapeutic blood flow. While respecting native cardiac flows, this support allows a significant reduction in its consumption, up to five times smaller than that observed in the Cardiac Assistance Devices (DAVG) currently on the market.
About the In-Vivo Study
The TET system was implanted for seven days, with five temperature sensors located throughout the internal coil pocket. The inner coil diameter is 8cm at 0.8 cm thick (same dimensions as an internal defibrillator, a device implanted daily in the G8 countries).
Heating was monitored for seven days with a consumption profile corresponding to a system operating at full speed, under extreme consumption conditions, well above nominal modes. The study showed that the average heating increase was around 1.5 +/- 0.2 °C, resulting in no tissue damage and no infection.
“The results of this study are remarkable and demonstrate that this small, mini-invasive TET system is functional and compatible with FDA guidelines and current regulations,” said professor Michael Acker, M.D., head of the Cardiac Surgery at the University of Pennsylvania Medical Center and FineHeart scientific advisory board member. “The ICOMS technology’s unique ability to replicate the volumetric displacement of the blood, beat by beat, by generating physiological cardiac flows addresses a significant unmet clinical need and could improve the quality of life for a huge population suffering from severe heart failure today and tomorrow.”
Heart failure (H.F.) is the second leading cause of death in the U.S. and Europe, a global pandemic affecting at least 26 million people worldwide and increasing prevalence. It is a degenerative disease leading to poor quality of life, frequent, costly hospitalizations, and early mortality.