Mölnlycke12.01.20
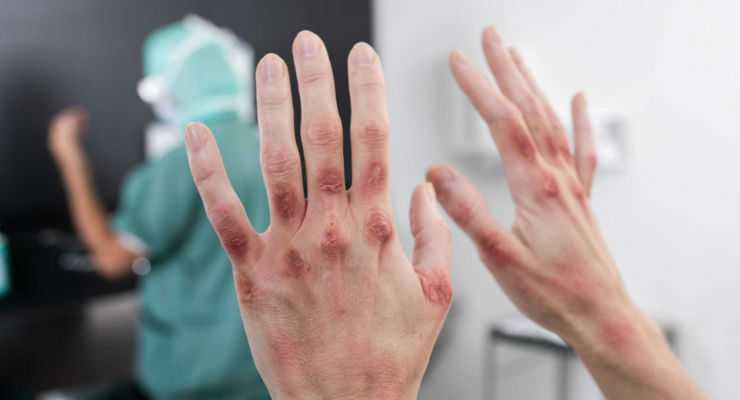
Mölnlycke has launched the Biogel PI UltraTouch S surgical glove, which addresses the problem of allergic contact dermatitis among surgical teams. Frequent handwashing and exposure to chemicals all contribute to occupational contact dermatitis. Exposure to chemical additives used in the manufacturing of gloves can cause irritant or allergic contact dermatitis. Almost one-third of surgeons and nurses surveyed indicate they suffered a skin reaction in the operating room, according to a recent survey.1
The latest addition to the Biogel product line, Biogel PI UltraTouch S surgical gloves have been designed with a new skin-friendly formula that is clinically proven to minimize the risk of allergic contact dermatitis in the operating room.* It is the first surgical glove to be cleared by the U.S. Food and Drug Administration (FDA) to reduce the potential for sensitizing users to chemical additives.
Precision Without Irritation
Offering the same precision, tactile sensitivity and perforation detection that has made Biogel the trusted favorite of healthcare professionals worldwide, Biogel PI UltraTouch S are made without the chemical accelerators known to cause allergic contact dermatitis*. Symptoms of allergic contact dermatitis include redness, cracked skin, sores, open lesions and scabbing.
The Scale of Contact Dermatitis
This year, a Mölnlycke-commissioned survey conducted by SERMO, the global network of healthcare professionals, revealed a shocking level of irritant and allergic contact dermatitis among surgeons and nurses working in the U.S. healthcare system:1
Instances of Type IV allergic contact dermatitis place surgeons and nurses at considerable harm and cause severe burdens on hospitals and health systems with lost productivity and occupational treatment costs. The cost of treating a single clinician with a glove-related allergy is estimated to be between $1,955 and $11,184 per year.2
Providing surgeons and their teams with dermatologically appropriate personal protective equipment (PPE) is the vital first step to ensuring a safe, effective and efficient operating room.
Commenting on the launch of Biogel PI UltraTouch S surgical gloves, Jeanne Marie Leahy, vice president of Surgical Marketing of Mölnlycke Health Care, said: “In the O.R., precision can be the difference between life and death. We engineered these new gloves to not only protect clinicians’ hands, but also to support their precision. It seems fitting to [have] a new way to improve working conditions for surgical team members who suffer from the pain and discomfort of allergic contact dermatitis. Contact dermatitis has long been a distraction to surgeons and nurses in the operating room. With Biogel PI UltraTouch S, these medical professionals now have a high-performing glove that allows them to do the very best job free from worry about experiencing skin reactions from chemical accelerators. Surgical team members suffering from allergic contact dermatitis can now enjoy the same precision, fit and safety of other Biogel surgical gloves.”
Biogel PI UltraTouch S surgical gloves are available now to hospitals across the United States.
Biogel PI UltraTouch S has received FDA 510k clearance for reduced potential for sensitizing users to chemical additives, the first sterile surgical glove to be awarded this claim. Modified Draize-95 patch testing was completed on 200 participants.
* Manufactured without the chemical accelerators known to cause irritant or Type IV allergic contact dermatitis including Dithiocarbamate (DTC), Diphenyl thiourea (DPTU), Diphenylguanidine (DPG), Zinc mercaptobenzothiazole (ZMBT), Thiurams
References
1 Global Surgeon and Nurse Survey conducted by SERMO, 2020. Data on File.
2 Politiek K. et.al. Systematic review of cost of-illness studies in hand eczema. Contact Dermatitis 2016.
The latest addition to the Biogel product line, Biogel PI UltraTouch S surgical gloves have been designed with a new skin-friendly formula that is clinically proven to minimize the risk of allergic contact dermatitis in the operating room.* It is the first surgical glove to be cleared by the U.S. Food and Drug Administration (FDA) to reduce the potential for sensitizing users to chemical additives.
Precision Without Irritation
Offering the same precision, tactile sensitivity and perforation detection that has made Biogel the trusted favorite of healthcare professionals worldwide, Biogel PI UltraTouch S are made without the chemical accelerators known to cause allergic contact dermatitis*. Symptoms of allergic contact dermatitis include redness, cracked skin, sores, open lesions and scabbing.
The Scale of Contact Dermatitis
This year, a Mölnlycke-commissioned survey conducted by SERMO, the global network of healthcare professionals, revealed a shocking level of irritant and allergic contact dermatitis among surgeons and nurses working in the U.S. healthcare system:1
- Thirty-one percent of surgeons and nurses surveyed had suffered from a skin reaction in the operating room
- Sixty percent of those had experienced a skin reaction in the last year
- Twenty-eight percent of surgeons had been forced to change gloves during a procedure due to a skin reaction
- Twenty-two percent had been distracted from work due to a skin reaction from surgical gloves
- Forty-nine percent had seen a colleague distracted due to a skin reaction from surgical gloves
Instances of Type IV allergic contact dermatitis place surgeons and nurses at considerable harm and cause severe burdens on hospitals and health systems with lost productivity and occupational treatment costs. The cost of treating a single clinician with a glove-related allergy is estimated to be between $1,955 and $11,184 per year.2
Providing surgeons and their teams with dermatologically appropriate personal protective equipment (PPE) is the vital first step to ensuring a safe, effective and efficient operating room.
Commenting on the launch of Biogel PI UltraTouch S surgical gloves, Jeanne Marie Leahy, vice president of Surgical Marketing of Mölnlycke Health Care, said: “In the O.R., precision can be the difference between life and death. We engineered these new gloves to not only protect clinicians’ hands, but also to support their precision. It seems fitting to [have] a new way to improve working conditions for surgical team members who suffer from the pain and discomfort of allergic contact dermatitis. Contact dermatitis has long been a distraction to surgeons and nurses in the operating room. With Biogel PI UltraTouch S, these medical professionals now have a high-performing glove that allows them to do the very best job free from worry about experiencing skin reactions from chemical accelerators. Surgical team members suffering from allergic contact dermatitis can now enjoy the same precision, fit and safety of other Biogel surgical gloves.”
Biogel PI UltraTouch S surgical gloves are available now to hospitals across the United States.
Biogel PI UltraTouch S has received FDA 510k clearance for reduced potential for sensitizing users to chemical additives, the first sterile surgical glove to be awarded this claim. Modified Draize-95 patch testing was completed on 200 participants.
* Manufactured without the chemical accelerators known to cause irritant or Type IV allergic contact dermatitis including Dithiocarbamate (DTC), Diphenyl thiourea (DPTU), Diphenylguanidine (DPG), Zinc mercaptobenzothiazole (ZMBT), Thiurams
References
1 Global Surgeon and Nurse Survey conducted by SERMO, 2020. Data on File.
2 Politiek K. et.al. Systematic review of cost of-illness studies in hand eczema. Contact Dermatitis 2016.