Globe Newswire09.21.20
Nu-Med Plus Inc., a medical device company which investigates and develops applications and devices for nitric oxide delivery in the medical field, announced that the U.S. Patent and Trademark Office has issued U.S. patent #10682486 for a single-use or short-term use nitric oxide inhaler.
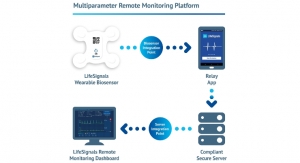
This patent will provide the company the intellectual property protection to pursue individual nitric oxide treatment options in emergency situations. The company has designed the system to be used in remote or rural areas which may not have access to non-portable equipment. The portable system has the capability to rapidly deliver the prescribed dose of nitric oxide when critical care and instantaneous use is required either within or outside of traditional hospital settings.
Typically, nitric oxide delivery devices are large, electronically complex devices that are used primarily in neonatal intensive care units. The importance of nitric oxide as a treatment is well documented in literature, but to date has not seen widespread use because of the bulky delivery equipment designed for hospital use and the high cost of the treatment.
Jeff Robins, CEO of Nu-Med Plus, stated, “This published patent is a major accomplishment for our company and can help open new patient markets. We believe that this patent will give our company the opportunity to be on the forefront of re-shaping the inhaled nitric oxide market landscape,”
Patent attorney of record is Bao Tran of Tran & Associates.
This patent will provide the company the intellectual property protection to pursue individual nitric oxide treatment options in emergency situations. The company has designed the system to be used in remote or rural areas which may not have access to non-portable equipment. The portable system has the capability to rapidly deliver the prescribed dose of nitric oxide when critical care and instantaneous use is required either within or outside of traditional hospital settings.
Typically, nitric oxide delivery devices are large, electronically complex devices that are used primarily in neonatal intensive care units. The importance of nitric oxide as a treatment is well documented in literature, but to date has not seen widespread use because of the bulky delivery equipment designed for hospital use and the high cost of the treatment.
Jeff Robins, CEO of Nu-Med Plus, stated, “This published patent is a major accomplishment for our company and can help open new patient markets. We believe that this patent will give our company the opportunity to be on the forefront of re-shaping the inhaled nitric oxide market landscape,”
Patent attorney of record is Bao Tran of Tran & Associates.