Globe Newswire09.14.20
According to Market Study Report LLC the global infectious disease test device market attained a valuation of $16.4 billion in 2019 and is projected to reach $26.8 billion by 2027, with a compound annual growth rate of 5.6 percent over 2020-2027. Increased pervasiveness of infectious diseases such as HPV, influenza, hepatitis, tuberculosis, and HIV is fueling the industry growth, cites the report. Moreover, extensive use of genomics and proteomics and molecular diagnostics technologies for infectious disease testing owing to rising patient awareness towards benefits of personalized medicines serves as a major impetus for the market growth.
In April 2015, Genome Canada, a funding source for genomics and proteomics, declared its plan for $16 million investment between April 2015 to March 2017. Besides this, NCBI stated that globally 17 million HIV-positive individuals are receiving life-extending drugs as compared to only 2.2 million people who were on antiretroviral therapy (ART) in 2005. Thus, inflow of investments towards development of new treatments and growing availability of testing kits across the globe is rendering a positive impact on global infectious disease test device market.
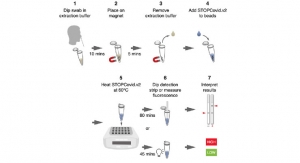
For the uninitiated, infectious disease diagnostics involves detecting presence of foreign antigen/organism by using specialized diagnostic kits. Majority of the standardized test kits available are designed for testing bacterial infections and sexually transmitted diseases. However, malaria, leptospirosis, dengue, and other diagnostic test are soon to be replaced by molecular techniques in the coming future.
Despite the positive outlook of the industry, unfavorable reimbursement scenario will hinder the market expansion. For instance, Medicare has reworked its reimbursement mechanism for some in-vitro diagnostic (IVD) tests, including molecular infectious disease tests. Some of these molecular pathology tests are charged using unlisted codes as they do not have their own HCPS (Healthcare Common Procedure Coding System), which negatively impacts the industry growth. Additionally, reimbursement for IVD tests is defined by the U.S. reimbursement system which only covers commercialized tests and ignores those primarily used for R&D applications, acting as major challenge for the industry.
On the geographical front, North America held a 44 percent share of global infectious disease test device market in 2019 and is expected to follow similar growth trend over the forecast duration. High awareness among healthcare professionals as well as patients, proactive government initiatives, and well-established healthcare infrastructure are supporting the regional industry expansion.
Meanwhile, Asia Pacific market is anticipated to expand at a rapid pace through 2027, set by favorable government initiatives for infectious disease testing, rising burden of unmet medical needs, improving economy and subsequent rise in disposable income. Furthermore, increased healthcare expenditure in emerging economies like India, China, and South Korea is boosting the industry growth in APAC.
In April 2015, Genome Canada, a funding source for genomics and proteomics, declared its plan for $16 million investment between April 2015 to March 2017. Besides this, NCBI stated that globally 17 million HIV-positive individuals are receiving life-extending drugs as compared to only 2.2 million people who were on antiretroviral therapy (ART) in 2005. Thus, inflow of investments towards development of new treatments and growing availability of testing kits across the globe is rendering a positive impact on global infectious disease test device market.
For the uninitiated, infectious disease diagnostics involves detecting presence of foreign antigen/organism by using specialized diagnostic kits. Majority of the standardized test kits available are designed for testing bacterial infections and sexually transmitted diseases. However, malaria, leptospirosis, dengue, and other diagnostic test are soon to be replaced by molecular techniques in the coming future.
Despite the positive outlook of the industry, unfavorable reimbursement scenario will hinder the market expansion. For instance, Medicare has reworked its reimbursement mechanism for some in-vitro diagnostic (IVD) tests, including molecular infectious disease tests. Some of these molecular pathology tests are charged using unlisted codes as they do not have their own HCPS (Healthcare Common Procedure Coding System), which negatively impacts the industry growth. Additionally, reimbursement for IVD tests is defined by the U.S. reimbursement system which only covers commercialized tests and ignores those primarily used for R&D applications, acting as major challenge for the industry.
On the geographical front, North America held a 44 percent share of global infectious disease test device market in 2019 and is expected to follow similar growth trend over the forecast duration. High awareness among healthcare professionals as well as patients, proactive government initiatives, and well-established healthcare infrastructure are supporting the regional industry expansion.
Meanwhile, Asia Pacific market is anticipated to expand at a rapid pace through 2027, set by favorable government initiatives for infectious disease testing, rising burden of unmet medical needs, improving economy and subsequent rise in disposable income. Furthermore, increased healthcare expenditure in emerging economies like India, China, and South Korea is boosting the industry growth in APAC.