Globe Newswire07.06.20
Michigan-based medical manufacturer and distributor, Alliant Healthcare Products, has expanded its product offerings to include COVID-19 IgG/IgM Rapid Test Kits.
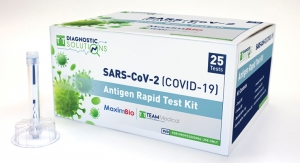
According to the company, the Premiere Bio-tech Rapid Test Kit “meets the highest degree of accuracy, availability, and affordability on the market” and sells at $18.50 per test. It has also been granted FDA Emergency Use Authorization. The kit, which offers results in 10 minutes, allows individuals to screen for both IgG and IgM strands of the coronavirus in 12 steps. In an effort to ensure those who are ready to return to work are able to do so safely, Alliant has made the kits available for purchase through its sister company, MediSurge.
The test detects IgM and IgG antibodies by analyzing the blood drawn from the user’s finger prick. IgM antibodies are typically associated with the early onset phase of the infection, while IgG is often associated with long-term immunity.
“We are proud to continue to expand our product offerings to meet the needs of our customers and our community,” said Bob Taylor, CEO of Alliant Healthcare. “We want to continue to invest in products that will help us to advance as a nation. Each test kit is designed to help ease the stress and worry of our frontline workers, healthcare organizations, government agencies, hospitals, businesses, families, and more.”
How It Works
Users of the kit are first prompted to disinfect the area they intend to prick for the test (fingertip) with an alcohol swab provided within the kit. After disinfection, users are instructed to use the sterile lancet to draw a small amount of blood from the targeted area. Using the disposable capillary, users should then deposit their blood sample into the test buffer marked (S) as well as add two drops to the buffer marked (B). After roughly 10 minutes, results for the test should begin to appear. Full test kit instructions have been made available here.
Results from the test kit can vary. Patients who test positive for RNA, but negative for both IgM and IgG strands may be in the window period of the infection. Those who test positive for RNA and IgM but negative for the IgG strand may be in the early stages of infection. Test results that are positive for RNA, IgM, and IgG strands are those in the active phase of infection. Patients that test positive for RNA, and IgG but negative for IgM are in the recurrent stage of infection. Results negative for RNA and IgM but positive for IgG are patients who are past infection and in the recovery stage.
The COVID-19 IgG/IgM test has been evaluated under 113 blood samples, including patients with preexisting conditions like pneumonia or respiratory symptoms. Individual kits are being sold at a marking of $18.50 per test, while large cases of 100 or more are sold at a reduced rate of $18.00 per test.
According to the company, the Premiere Bio-tech Rapid Test Kit “meets the highest degree of accuracy, availability, and affordability on the market” and sells at $18.50 per test. It has also been granted FDA Emergency Use Authorization. The kit, which offers results in 10 minutes, allows individuals to screen for both IgG and IgM strands of the coronavirus in 12 steps. In an effort to ensure those who are ready to return to work are able to do so safely, Alliant has made the kits available for purchase through its sister company, MediSurge.
The test detects IgM and IgG antibodies by analyzing the blood drawn from the user’s finger prick. IgM antibodies are typically associated with the early onset phase of the infection, while IgG is often associated with long-term immunity.
“We are proud to continue to expand our product offerings to meet the needs of our customers and our community,” said Bob Taylor, CEO of Alliant Healthcare. “We want to continue to invest in products that will help us to advance as a nation. Each test kit is designed to help ease the stress and worry of our frontline workers, healthcare organizations, government agencies, hospitals, businesses, families, and more.”
How It Works
Users of the kit are first prompted to disinfect the area they intend to prick for the test (fingertip) with an alcohol swab provided within the kit. After disinfection, users are instructed to use the sterile lancet to draw a small amount of blood from the targeted area. Using the disposable capillary, users should then deposit their blood sample into the test buffer marked (S) as well as add two drops to the buffer marked (B). After roughly 10 minutes, results for the test should begin to appear. Full test kit instructions have been made available here.
Results from the test kit can vary. Patients who test positive for RNA, but negative for both IgM and IgG strands may be in the window period of the infection. Those who test positive for RNA and IgM but negative for the IgG strand may be in the early stages of infection. Test results that are positive for RNA, IgM, and IgG strands are those in the active phase of infection. Patients that test positive for RNA, and IgG but negative for IgM are in the recurrent stage of infection. Results negative for RNA and IgM but positive for IgG are patients who are past infection and in the recovery stage.
The COVID-19 IgG/IgM test has been evaluated under 113 blood samples, including patients with preexisting conditions like pneumonia or respiratory symptoms. Individual kits are being sold at a marking of $18.50 per test, while large cases of 100 or more are sold at a reduced rate of $18.00 per test.