GlobeNewswire06.17.20
Fulgent Genetics Inc., a provider of comprehensive genetic testing solutions, has launched an at-home testing solution for the Coronavirus Disease (COVID-19). The company has received Emergency Use Authorization (EUA) from the US Food and Drug Administration (FDA) for use in the United States.
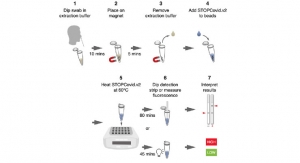
Fulgent Genetics will be offering the testing service through Picture Genetics, the company’s consumer-initiated genetic testing platform, beginning next week. Individuals interested in ordering testing services must qualify for the service through an online eligibility screener, which makes eligibility determinations based on current CDC guidelines while prioritizing those who are most in need of testing. Eligible individuals will receive Fulgent’s collection materials in the mail that enable them to self-collect their specimen with a mid-turbinate nasal swab, which will then be sent back to Fulgent’s CLIA-certified high-complexity, CAP-accredited laboratory in Temple City, Calif. By enabling individuals to collect samples themselves, Fulgent’s at-home test services conserve valuable time and PPE resources otherwise required to perform these testing services for patients. Patients will receive their results through the Picture Genetics platform within 24-48 hours from receipt of the sample.
Fulgent has partnered with a national clinician network on its at-home COVID-19 test which will oversee the process including approval of each test ordered, monitoring of materials produced, and review of reports provided for patients by Fulgent Genetics after testing is completed.
“We are pleased to be able to offer a convenient and readily available COVID-19 testing solution for individuals at risk of contracting the virus,” said Brandon Perthuis, Chief Commercial Officer at Fulgent Genetics. “As the coronavirus continues to spread, options for testing remain limited and many eligible individuals are unable to get the testing they need. We believe our at-home testing service will both enable at-risk individuals, particularly those at the front lines of this pandemic, to more readily access testing solutions, while potentially offering a solution for organizations to screen employees as they return to work. We believe that increasing availability of at-home testing solutions in general will help to alleviate congestion at health care facilities, reduce contribution to more community spread, and offer individuals who are unable to leave their homes a safer and more convenient testing solution,” further commented Perthuis. “Since launching our RT-PCR Test (non at-home test) several months ago, we have won several strategic accounts and are processing thousands of tests daily with an average turnaround time of 24 hours from receipt of sample. This at-home COVID-19 test will diversify our go-to-market approach for tests by utilizing our existing consumer-initiated genetic testing platform.”
Fulgent Genetics’ at-home test service for COVID-19 has been granted an EUA by the FDA only for the detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens.
Fulgent Genetics will be offering the testing service through Picture Genetics, the company’s consumer-initiated genetic testing platform, beginning next week. Individuals interested in ordering testing services must qualify for the service through an online eligibility screener, which makes eligibility determinations based on current CDC guidelines while prioritizing those who are most in need of testing. Eligible individuals will receive Fulgent’s collection materials in the mail that enable them to self-collect their specimen with a mid-turbinate nasal swab, which will then be sent back to Fulgent’s CLIA-certified high-complexity, CAP-accredited laboratory in Temple City, Calif. By enabling individuals to collect samples themselves, Fulgent’s at-home test services conserve valuable time and PPE resources otherwise required to perform these testing services for patients. Patients will receive their results through the Picture Genetics platform within 24-48 hours from receipt of the sample.
Fulgent has partnered with a national clinician network on its at-home COVID-19 test which will oversee the process including approval of each test ordered, monitoring of materials produced, and review of reports provided for patients by Fulgent Genetics after testing is completed.
“We are pleased to be able to offer a convenient and readily available COVID-19 testing solution for individuals at risk of contracting the virus,” said Brandon Perthuis, Chief Commercial Officer at Fulgent Genetics. “As the coronavirus continues to spread, options for testing remain limited and many eligible individuals are unable to get the testing they need. We believe our at-home testing service will both enable at-risk individuals, particularly those at the front lines of this pandemic, to more readily access testing solutions, while potentially offering a solution for organizations to screen employees as they return to work. We believe that increasing availability of at-home testing solutions in general will help to alleviate congestion at health care facilities, reduce contribution to more community spread, and offer individuals who are unable to leave their homes a safer and more convenient testing solution,” further commented Perthuis. “Since launching our RT-PCR Test (non at-home test) several months ago, we have won several strategic accounts and are processing thousands of tests daily with an average turnaround time of 24 hours from receipt of sample. This at-home COVID-19 test will diversify our go-to-market approach for tests by utilizing our existing consumer-initiated genetic testing platform.”
Fulgent Genetics’ at-home test service for COVID-19 has been granted an EUA by the FDA only for the detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens.