GlobeNewswire06.15.20
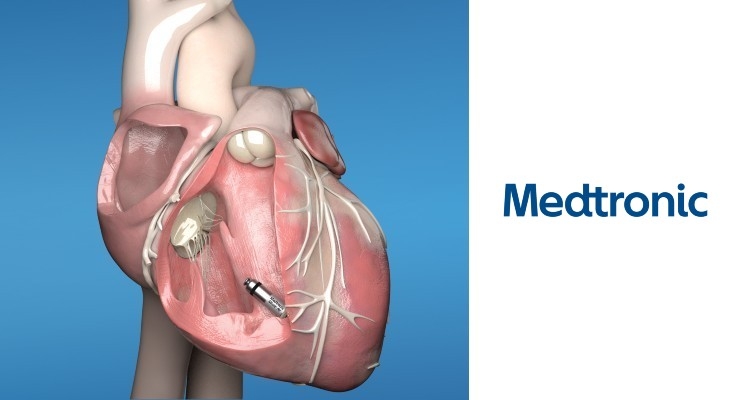
Medtronic plc received CE mark for Micra AV Transcatheter Pacing System (TPS), the world’s smallest pacemaker with atrioventricular (AV) synchrony. Micra AV is indicated for the treatment of patients with AV block, a condition in which the electrical signals between the chambers of the heart (the atria and the ventricle) are impaired. Medtronic now offers the first and only CE Mark approved leadless pacemaker portfolio, expanding the number of potential candidates for this groundbreaking technology throughout the world.
First implants of Micra AV recently occurred in Spain by Dr. José Ramón González Juanatey at the University Hospital in Santiago de Compostela.
"This new device not only stimulates but is also able to recognize the electrical activity of the whole heart. Our ultimate goal is to bring the latest cardiovascular innovation to patients in our area. Now we can extend this wireless technology to other patients who require dual chamber stimulation and in whom traditional stimulation cannot be performed or is conditioned by previous infections, occlusions of the vessels of the upper extremities, etc.," said Dr. José Ramón González Juanatey, director, Cardiology and Intensive Cardiac Care Department, full professor of Cardiology, University Hospital, Santiago de Compostela, Spain (past-president, Spanish Society of Cardiology).
Historically, patients with AV block have been treated with traditional dual-chamber pacemakers which are implanted in the upper chest, under the skin below the collar bone, and connected to the heart using thin wires called “leads.” Identical in size and shape to the original Micra TPS, Micra AV has several additional internal atrial sensing algorithms which detect cardiac movement, allowing the device to adjust pacing in the ventricle to coordinate with the atrium, providing “AV synchronous” pacing therapy to patients with AV block.
The Micra AV approval is based on data from the MARVEL 2 (Micra Atrial Tracking Using A Ventricular accELerometer) study, which evaluated the safety and effectiveness of accelerometer-based atrial sensing algorithms. The study evaluated the ability of the Micra’s internal sensor to monitor and detect atrial contractions and enable coordinated pacing between the atrium and ventricle, thereby providing AV synchrony. Results from the study, presented at the American Heart Association 2019 Scientific Sessions and published simultaneously in JACC: Clinical Electrophysiology, showed the primary efficacy objective was met, with a significantly greater percentage of complete heart block patients with normal sinus rhythm having >70% AV synchrony during algorithm-mediated AV synchronous pacing (38 of 40 patients, 95%) than VVI pacing (0 patients, P<0.001 for proportion of patients with >70% synchrony). The study’s primary safety objective was also met, with no pauses or episodes of pacing-induced tachycardia reported during algorithm mediated AV synchronous pacing.
“From the first battery-operated external pacemaker in 1957 to the innovative Micra leadless pacemaker portfolio, Medtronic continues to provide pioneering pacing solutions to physicians and their patients,” said Alphons Vincent, M.D., medical director for Europe, the Middle East & Africa of the Cardiac Rhythm and Heart Failure division, which is part of the Cardiac and Vascular Group at Medtronic.
Micra AV won FDA approval in January. Find out more here
First implants of Micra AV recently occurred in Spain by Dr. José Ramón González Juanatey at the University Hospital in Santiago de Compostela.
"This new device not only stimulates but is also able to recognize the electrical activity of the whole heart. Our ultimate goal is to bring the latest cardiovascular innovation to patients in our area. Now we can extend this wireless technology to other patients who require dual chamber stimulation and in whom traditional stimulation cannot be performed or is conditioned by previous infections, occlusions of the vessels of the upper extremities, etc.," said Dr. José Ramón González Juanatey, director, Cardiology and Intensive Cardiac Care Department, full professor of Cardiology, University Hospital, Santiago de Compostela, Spain (past-president, Spanish Society of Cardiology).
Historically, patients with AV block have been treated with traditional dual-chamber pacemakers which are implanted in the upper chest, under the skin below the collar bone, and connected to the heart using thin wires called “leads.” Identical in size and shape to the original Micra TPS, Micra AV has several additional internal atrial sensing algorithms which detect cardiac movement, allowing the device to adjust pacing in the ventricle to coordinate with the atrium, providing “AV synchronous” pacing therapy to patients with AV block.
The Micra AV approval is based on data from the MARVEL 2 (Micra Atrial Tracking Using A Ventricular accELerometer) study, which evaluated the safety and effectiveness of accelerometer-based atrial sensing algorithms. The study evaluated the ability of the Micra’s internal sensor to monitor and detect atrial contractions and enable coordinated pacing between the atrium and ventricle, thereby providing AV synchrony. Results from the study, presented at the American Heart Association 2019 Scientific Sessions and published simultaneously in JACC: Clinical Electrophysiology, showed the primary efficacy objective was met, with a significantly greater percentage of complete heart block patients with normal sinus rhythm having >70% AV synchrony during algorithm-mediated AV synchronous pacing (38 of 40 patients, 95%) than VVI pacing (0 patients, P<0.001 for proportion of patients with >70% synchrony). The study’s primary safety objective was also met, with no pauses or episodes of pacing-induced tachycardia reported during algorithm mediated AV synchronous pacing.
“From the first battery-operated external pacemaker in 1957 to the innovative Micra leadless pacemaker portfolio, Medtronic continues to provide pioneering pacing solutions to physicians and their patients,” said Alphons Vincent, M.D., medical director for Europe, the Middle East & Africa of the Cardiac Rhythm and Heart Failure division, which is part of the Cardiac and Vascular Group at Medtronic.
Micra AV won FDA approval in January. Find out more here