Business Wire05.13.20
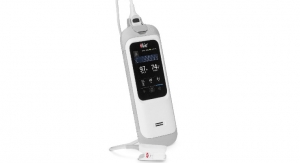
Mauna Kea Technologies, inventor of Cellvizio, the multidisciplinary probe- and needle-based confocal laser endomicroscopy (pCLE/nCLE) platform, has obtained 510(k) clearance from the U.S. Food and Drug Administration (FDA) and CE marking of the next-generation Cellvizio endomicroscopy platform, built with the company’s new proprietary system architecture. This marks the 18th FDA 510(k) clearance of the Cellvizio pCLE/nCLE platform.
The new Cellvizio incorporates breakthrough modular design solutions to facilitate and better integrate endomicroscopy within procedure suites as well as within third-party platforms. The new platform’s hardware and software design was built from the ground up to facilitate future developments, including integration of deep learning (artificial intelligence) capabilities for assisted image interpretation. The ergonomic and significantly reduced footprint, integrates easily with laparoscopic, advanced navigation, and robotic systems. This novel platform is also capable of hosting other proprietary endomicroscopic architectures with imaging capabilities at other wavelengths supporting fluorescence-guided surgery and molecular imaging.
With a completely redesigned user interface, Cellvizio delivers smart workflows for greater efficiency and usability. The all-new touchscreen and single-handed probe connection provide for easier setup and use. Developed to bring precision imaging to more patients with nine dedicated Confocal Miniprobes, the new Cellvizio delivers premium imaging capabilities, offering clinicians an efficient high-performing, imaging solution for endoscopy, interventional, and surgical operating suites.
"We are proud to announce the FDA clearance and CE marking of our next generation Cellvizio in the USA and in Europe," said Robert L. Gershon, CEO of Mauna Kea Technologies. "The next generation Cellvizio platform will be launched commercially in the coming months and, with its modular and flexible design as well as being AI and molecular imaging ready, will host our future innovations, indications and support partnership developments."
In January, Mauna Kea Technologies received FDA 510(k) clearance to market the Cellvizio 100 Series and all confocal mini-probes combined with a fluorescent marker, fluorescein, for the additional indication of visualization of blood flow.
The Cellvizio 100 Series and all of its probes make it possible to visualize the internal microstructure of tissues, but also (and not only) to identify cells, vessels and their organization or architecture. The endomicroscopy system is used in gastroenterology, urology and pneumology during endoscopic, laparoscopic (manual and robot-assisted), and percutaneous imaging-guided procedures.
The new Cellvizio incorporates breakthrough modular design solutions to facilitate and better integrate endomicroscopy within procedure suites as well as within third-party platforms. The new platform’s hardware and software design was built from the ground up to facilitate future developments, including integration of deep learning (artificial intelligence) capabilities for assisted image interpretation. The ergonomic and significantly reduced footprint, integrates easily with laparoscopic, advanced navigation, and robotic systems. This novel platform is also capable of hosting other proprietary endomicroscopic architectures with imaging capabilities at other wavelengths supporting fluorescence-guided surgery and molecular imaging.
With a completely redesigned user interface, Cellvizio delivers smart workflows for greater efficiency and usability. The all-new touchscreen and single-handed probe connection provide for easier setup and use. Developed to bring precision imaging to more patients with nine dedicated Confocal Miniprobes, the new Cellvizio delivers premium imaging capabilities, offering clinicians an efficient high-performing, imaging solution for endoscopy, interventional, and surgical operating suites.
"We are proud to announce the FDA clearance and CE marking of our next generation Cellvizio in the USA and in Europe," said Robert L. Gershon, CEO of Mauna Kea Technologies. "The next generation Cellvizio platform will be launched commercially in the coming months and, with its modular and flexible design as well as being AI and molecular imaging ready, will host our future innovations, indications and support partnership developments."
In January, Mauna Kea Technologies received FDA 510(k) clearance to market the Cellvizio 100 Series and all confocal mini-probes combined with a fluorescent marker, fluorescein, for the additional indication of visualization of blood flow.
The Cellvizio 100 Series and all of its probes make it possible to visualize the internal microstructure of tissues, but also (and not only) to identify cells, vessels and their organization or architecture. The endomicroscopy system is used in gastroenterology, urology and pneumology during endoscopic, laparoscopic (manual and robot-assisted), and percutaneous imaging-guided procedures.