PR Newswire04.20.20
ELITechGroup MDx LLC, a global provider of diagnostic testing products, and its Korean partner, OSANG Healthcare, have received U.S. Food and Drug Administration (FDA) Emergency Use Authorization (EUA) for a SARS-CoV-2 virus test for use in the United States.
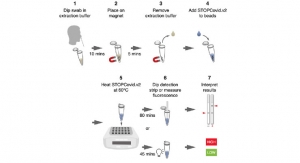
The GeneFinder COVID-19 Plus RealAmp Kit provides a fast and easy-to-use diagnostic solution to rapidly identify clusters of infection and has been validated using the ABI 7500 thermocycler and Bio-Rad CFX96 cycler—both popular Polymerase Chain Reaction (PCR) instruments found in diagnostics laboratories throughout the country.
Delivering qualitative results within hours, the highly sensitive testing kit requires a single tube rather than multiple reactions and uses Aveolar fluid, throat swab or sputum samples in a PCR instrument to detect the virus.
"The dramatic explosion of the novel coronavirus is affecting lives in every state," said ELITechGroup CEO Christoph Gauer. "Hospitals and labs need access to reliable COVID-19 test kits, and we are here to support the medical professionals on the front lines working to prevent the spread of COVID-19."
To date, ELITechGroup has delivered more than 700,000 COVID-19 CE-IVD tests worldwide that have proven effective in accurately detecting the infection. ELITechGroup MDx is the exclusive distributor of the test kits to certain markets across the United States.
"We are determined to fight COVID-19 and the alarming infection rates now being seen in the U.S.," said OSANG Healthcare Chairman Dong-Hyun Lee. "We are pleased to partner with ELITechGroup MDx to bring much needed testing kits to U.S. shores."
The authorization follows last month's launch of the Aerospray Cytology Slide Stainer Cytocentrifuge by ELITechGroup MDx.
The ELITechGroup’s Aerospray Cytology Slide Stainer/Cytocentrifuge automates the preparation and staining of liquid based cytology specimens to facilitate the examination of an array of cell differentiations utilizing Papanicolaou staining techniques.
The advantages of the Aerospray Cytology Slide Stainer/Cytocentrifuge is that the device performs two functions of sample preparation through cytocentrifugation and sample staining. The system maximizes cell recovery to the slide through the use of its Cytopro Rotor, eliminates cross-contamination with an application of fresh stain for each slide, and provides programmable regressive and progressive staining capabilities that can be stored in memory to meet the needs and preferences of any end user. The instrument standardizes staining across multiple labs, provides consistently high-quality control, is low maintenance, reduces the labor/cost per slide, enables laboratory compliance, and is the perfect back-up for high automation laboratories for specialty staining and during maintenance.
The GeneFinder COVID-19 Plus RealAmp Kit provides a fast and easy-to-use diagnostic solution to rapidly identify clusters of infection and has been validated using the ABI 7500 thermocycler and Bio-Rad CFX96 cycler—both popular Polymerase Chain Reaction (PCR) instruments found in diagnostics laboratories throughout the country.
Delivering qualitative results within hours, the highly sensitive testing kit requires a single tube rather than multiple reactions and uses Aveolar fluid, throat swab or sputum samples in a PCR instrument to detect the virus.
"The dramatic explosion of the novel coronavirus is affecting lives in every state," said ELITechGroup CEO Christoph Gauer. "Hospitals and labs need access to reliable COVID-19 test kits, and we are here to support the medical professionals on the front lines working to prevent the spread of COVID-19."
To date, ELITechGroup has delivered more than 700,000 COVID-19 CE-IVD tests worldwide that have proven effective in accurately detecting the infection. ELITechGroup MDx is the exclusive distributor of the test kits to certain markets across the United States.
"We are determined to fight COVID-19 and the alarming infection rates now being seen in the U.S.," said OSANG Healthcare Chairman Dong-Hyun Lee. "We are pleased to partner with ELITechGroup MDx to bring much needed testing kits to U.S. shores."
The authorization follows last month's launch of the Aerospray Cytology Slide Stainer Cytocentrifuge by ELITechGroup MDx.
The ELITechGroup’s Aerospray Cytology Slide Stainer/Cytocentrifuge automates the preparation and staining of liquid based cytology specimens to facilitate the examination of an array of cell differentiations utilizing Papanicolaou staining techniques.
The advantages of the Aerospray Cytology Slide Stainer/Cytocentrifuge is that the device performs two functions of sample preparation through cytocentrifugation and sample staining. The system maximizes cell recovery to the slide through the use of its Cytopro Rotor, eliminates cross-contamination with an application of fresh stain for each slide, and provides programmable regressive and progressive staining capabilities that can be stored in memory to meet the needs and preferences of any end user. The instrument standardizes staining across multiple labs, provides consistently high-quality control, is low maintenance, reduces the labor/cost per slide, enables laboratory compliance, and is the perfect back-up for high automation laboratories for specialty staining and during maintenance.