GlobeNewswire03.06.20
Neuronetics Inc., a commercial stage medical technology company focused on designing, developing, and marketing products that improve the quality of life for patients who suffer from psychiatric disorders, announced the U.S. Food and Drug Administration (FDA) has recently granted Breakthrough Device Designation for the company’s NeuroStar Advanced Therapy System for the treatment of bipolar depression.
The FDA’s Breakthrough Device Program is intended to help patients and healthcare providers receive more timely access to breakthrough technologies that have the potential to provide more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. The program creates an expedited pathway for prioritized FDA review of the NeuroStar Advanced Therapy clinical trial program. Neuronetics has submitted initial clinical plans for discussion with the FDA.
“We are delighted that the FDA has accepted our application for Breakthrough Device Designation and look forward to working closely with them to provide a potential solution for people with bipolar depression. This potential new treatment indication for NeuroStar means that millions of people with bipolar disorder who do not respond to medications may have a proven, non-drug treatment available to them,” said Yelena Tropsha, Vice President, Commercial Access. “The Breakthrough Device Designation is an important milestone that further supports our mission of renewing lives by transforming neurohealth.”
Bipolar I and II disorder cause recurrent, dramatic shifts in mood, energy, and activity levels. Bipolar disorder affects approximately 6.5-7 million U.S. adults annually.1,2 Focus for therapies has been on treating the mania and hypomania phase of the disorder with only a few medications approved for the depression phase associated with these disorders. With the Breakthrough Device Designation, NeuroStar Advanced Therapy—the market leader in transcranial magnetic stimulation (TMS) for unipolar major depression—could potentially provide more effective treatment with limited safety concerns to this additional patient population.
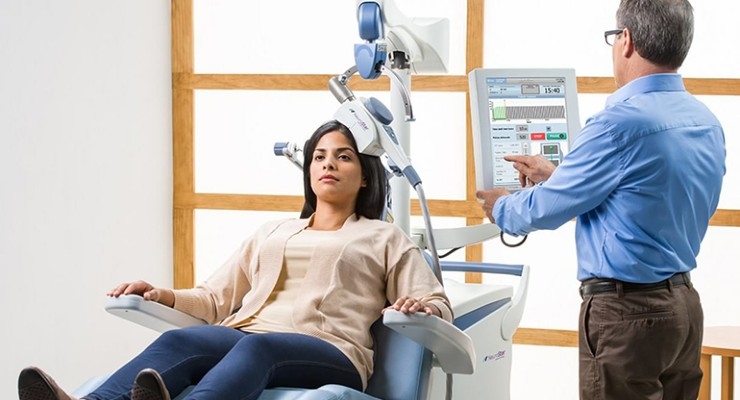
In 2008, NeuroStar Advanced Therapy was the first TMS device to receive FDA clearance for adults with Major Depressive Disorder who have not seen success with at least one antidepressant medication. NeuroStar is a non-invasive form of neuromodulation that uses magnetic pulses to stimulate areas of the brain that are underactive in depression.3,4 It is now the first TMS device to receive Breakthrough Device Designation for bipolar depression in adult patients with Bipolar I or Bipolar II disorders that have failed to receive satisfactory improvement from prior pharmacological therapy.
References
1 https://biomedtracker.com/indicationreport.cfm?indid=168 (Pharma Intelligence BioMed Tracker) based on U.S. adult population, Census July 2018
2 National Institute of Mental Health https://www.nimh.nih.gov/health/statistics/bipolar-disorders.html
3 Post A, et al. J Psychiatric Research, (2001)
4 Liston C, et al. Biol Psychiatry, (2014)
The FDA’s Breakthrough Device Program is intended to help patients and healthcare providers receive more timely access to breakthrough technologies that have the potential to provide more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. The program creates an expedited pathway for prioritized FDA review of the NeuroStar Advanced Therapy clinical trial program. Neuronetics has submitted initial clinical plans for discussion with the FDA.
“We are delighted that the FDA has accepted our application for Breakthrough Device Designation and look forward to working closely with them to provide a potential solution for people with bipolar depression. This potential new treatment indication for NeuroStar means that millions of people with bipolar disorder who do not respond to medications may have a proven, non-drug treatment available to them,” said Yelena Tropsha, Vice President, Commercial Access. “The Breakthrough Device Designation is an important milestone that further supports our mission of renewing lives by transforming neurohealth.”
Bipolar I and II disorder cause recurrent, dramatic shifts in mood, energy, and activity levels. Bipolar disorder affects approximately 6.5-7 million U.S. adults annually.1,2 Focus for therapies has been on treating the mania and hypomania phase of the disorder with only a few medications approved for the depression phase associated with these disorders. With the Breakthrough Device Designation, NeuroStar Advanced Therapy—the market leader in transcranial magnetic stimulation (TMS) for unipolar major depression—could potentially provide more effective treatment with limited safety concerns to this additional patient population.
In 2008, NeuroStar Advanced Therapy was the first TMS device to receive FDA clearance for adults with Major Depressive Disorder who have not seen success with at least one antidepressant medication. NeuroStar is a non-invasive form of neuromodulation that uses magnetic pulses to stimulate areas of the brain that are underactive in depression.3,4 It is now the first TMS device to receive Breakthrough Device Designation for bipolar depression in adult patients with Bipolar I or Bipolar II disorders that have failed to receive satisfactory improvement from prior pharmacological therapy.
References
1 https://biomedtracker.com/indicationreport.cfm?indid=168 (Pharma Intelligence BioMed Tracker) based on U.S. adult population, Census July 2018
2 National Institute of Mental Health https://www.nimh.nih.gov/health/statistics/bipolar-disorders.html
3 Post A, et al. J Psychiatric Research, (2001)
4 Liston C, et al. Biol Psychiatry, (2014)