Robocath03.02.20
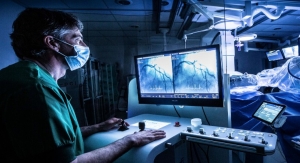
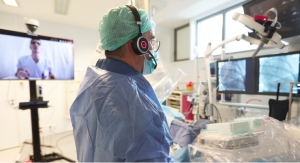
Robocath, a company that designs, develops and commercializes cardiovascular robotic systems for the treatment of vascular diseases, has successfully completed its first robotic coronary angioplasties with R-One in Germany. The percutaneous coronary interventions (PCI) were performed by Pr. Michael Haude, a recognized and highly experienced interventional cardiologist at Rheinland Klinikum Neuss Lukaskrankenhaus, and his team.
Haude, director of Medical Clinic I at Rheinland Klinikum Neuss Lukaskrankenhaus, said, “I’m proud to be the first user of this brand new European robotic solution for PCI in Germany. These robotic interventions pave the way for a new era in interventional cardiology. The benefits brought by R-One are huge for practitioners and will drastically improve our working conditions, which will consequently benefit patient care. My team and I look forward to integrating this robotic-assisted platform for PCI into our day-to-day clinical practice.”
Philippe Bencteux, chairman and founder of Robocath, added, “We are delighted with the success of these procedures and would like to thank Pr. Haude’s team for their enthusiasm. Pr. Haude has over 30 years’ experience in interventional cardiology and we rely on capitalizing on this kind of experience to develop clinical evidence and demonstrate all the benefits of our solution.”
R-One is the first solution developed by Robocath. It uses a unique technology that optimizes the safety of robotic-assisted coronary angioplasty. This medical procedure consists of revascularizing the cardiac muscle by inserting one or more implants (stents) into the arteries that supply it with blood. Every 30 seconds, somewhere in the world, this type of procedure is performed.
R-One is designed to operate with precision and perform specific movements, creating better interventional conditions. Thanks to its open architecture, R-One is compatible with market-leading devices and cath labs. It received the CE marking in February 2019 and started its clinical application in September 2019.
Lucien Goffart, CEO of Robocath, said, “These first procedures in Germany are in line with our expansion strategy. We are very proud to achieve this important strategic step and to see our first robot used in this key market. There is a momentum for vascular robotics today as it completely addresses the medico-economic challenges of modern healthcare systems. The interest of major industry players is a clear indicator of this; it demonstrates that the penetration of robotics in the medical sector is now irreversible."
Robocath performed its first robotic coronary angioplasties with R-One in Africa last December.
Haude, director of Medical Clinic I at Rheinland Klinikum Neuss Lukaskrankenhaus, said, “I’m proud to be the first user of this brand new European robotic solution for PCI in Germany. These robotic interventions pave the way for a new era in interventional cardiology. The benefits brought by R-One are huge for practitioners and will drastically improve our working conditions, which will consequently benefit patient care. My team and I look forward to integrating this robotic-assisted platform for PCI into our day-to-day clinical practice.”
Philippe Bencteux, chairman and founder of Robocath, added, “We are delighted with the success of these procedures and would like to thank Pr. Haude’s team for their enthusiasm. Pr. Haude has over 30 years’ experience in interventional cardiology and we rely on capitalizing on this kind of experience to develop clinical evidence and demonstrate all the benefits of our solution.”
R-One is the first solution developed by Robocath. It uses a unique technology that optimizes the safety of robotic-assisted coronary angioplasty. This medical procedure consists of revascularizing the cardiac muscle by inserting one or more implants (stents) into the arteries that supply it with blood. Every 30 seconds, somewhere in the world, this type of procedure is performed.
R-One is designed to operate with precision and perform specific movements, creating better interventional conditions. Thanks to its open architecture, R-One is compatible with market-leading devices and cath labs. It received the CE marking in February 2019 and started its clinical application in September 2019.
Lucien Goffart, CEO of Robocath, said, “These first procedures in Germany are in line with our expansion strategy. We are very proud to achieve this important strategic step and to see our first robot used in this key market. There is a momentum for vascular robotics today as it completely addresses the medico-economic challenges of modern healthcare systems. The interest of major industry players is a clear indicator of this; it demonstrates that the penetration of robotics in the medical sector is now irreversible."
Robocath performed its first robotic coronary angioplasties with R-One in Africa last December.