Business Wire02.19.20
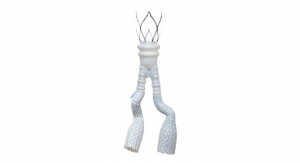
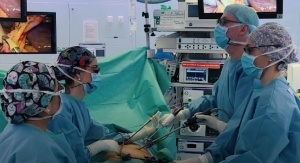
Silicon Valley-based medical device company PQ Bypass announced the 100th patient in the DETOUR2 Clinical Trial, which evaluates the safety and effectiveness of the minimally invasive DETOUR procedure for percutaneous femoropopliteal bypass. The case was performed by Dr. Jonathan Bowman at The Vascular Experts in Darien, Conn., the largest group of board-certified vascular surgeons in the United States. The treated TASC D lesion was 35 centimeters in length and included a 25-centimeter chronic total occlusion.
“We are thrilled to be a part of bringing a complex procedure out of the inpatient setting,” said Paul J. Gagne M.D., FACS, RVT, partner, The Vascular Experts, and member of the Outpatient Endovascular and Interventional Society. “In the past, surgical fem-pop bypass required at least a three-day inpatient stay with significant morbidity. With the advent of the PQ Bypass DETOUR system and this important research we are undertaking, we are able to send patients home the same day with just a couple of small incisions.”
“Our mission is to give physicians, and the patients they treat, another option in the treatment armamentarium for SFA lesions, which in our DETOUR 1 and DETOUR2 studies have averaged more than 30 centimeters in length,” said Heather Simonsen, general manager of PQ Bypass. “The 100th patient in the DETOUR2 clinical trial is a significant achievement on our path to address an unmet need in the treatment of advanced peripheral arterial disease.”
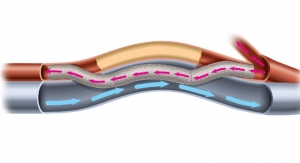
The 100th patient marks nearly 50 percent completion in the prospective, single-arm, global multicenter study, which will continue enrolling across the United States and Europe in 2020. Pending U.S. Food and Drug Administration (FDA) approval, the company aims to bridge the gap between the treat-and-repeat cycle of traditional endovascular therapies, and the current durability gold standard of bypass surgery, which some patients may be unwilling or unable to receive.
In partnership with New York-based Clinical Research Organization, Syntactx LLC, and its vascular experts Kenneth Ouriel M.D., president and CEO, and Mark Adelman M.D., chief medical officer, PQ Bypass is aggregating the pivotal trial data with the expectation of FDA approval and subsequent commercial distribution.
The DETOUR system, and TORUS Stent Graft are limited by U.S. law to investigational use only and is not available for sale.
“We are thrilled to be a part of bringing a complex procedure out of the inpatient setting,” said Paul J. Gagne M.D., FACS, RVT, partner, The Vascular Experts, and member of the Outpatient Endovascular and Interventional Society. “In the past, surgical fem-pop bypass required at least a three-day inpatient stay with significant morbidity. With the advent of the PQ Bypass DETOUR system and this important research we are undertaking, we are able to send patients home the same day with just a couple of small incisions.”
“Our mission is to give physicians, and the patients they treat, another option in the treatment armamentarium for SFA lesions, which in our DETOUR 1 and DETOUR2 studies have averaged more than 30 centimeters in length,” said Heather Simonsen, general manager of PQ Bypass. “The 100th patient in the DETOUR2 clinical trial is a significant achievement on our path to address an unmet need in the treatment of advanced peripheral arterial disease.”
The 100th patient marks nearly 50 percent completion in the prospective, single-arm, global multicenter study, which will continue enrolling across the United States and Europe in 2020. Pending U.S. Food and Drug Administration (FDA) approval, the company aims to bridge the gap between the treat-and-repeat cycle of traditional endovascular therapies, and the current durability gold standard of bypass surgery, which some patients may be unwilling or unable to receive.
In partnership with New York-based Clinical Research Organization, Syntactx LLC, and its vascular experts Kenneth Ouriel M.D., president and CEO, and Mark Adelman M.D., chief medical officer, PQ Bypass is aggregating the pivotal trial data with the expectation of FDA approval and subsequent commercial distribution.
The DETOUR system, and TORUS Stent Graft are limited by U.S. law to investigational use only and is not available for sale.