PR Newswire01.24.20
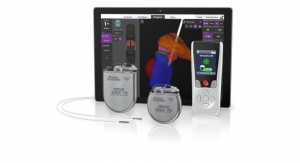
The expanded indication for Abbott's Infinity Deep Brain Stimulation (DBS) system includes targeting of the internal globus pallidus (GPi). The GPi plays an integral role in the motor function and can be targeted with DBS to improve the symptoms of Parkinson's not adequately controlled by medication.
According to Abbott, its Infinity DBS is now the only directional DBS system approved for all major targets used to treat movement disorders, Parkinson's disease, and Essential Tremor: the subthalamic nucleus (STN), ventral intermediate nucleus (VIM), and GPi. The DBS system operates in these indications on an iOS software platform with Bluetooth. Clinicians can streamline the programming process with an iPad mini device using Abbott's Informity Programming feature to achieve optimal outcomes with directional leads. Patients can discreetly manage symptoms with the system’s iPod touch controller.
"The internal segment of the global pallidus, or GPi, is a well-established valuable DBS target for the management of the motor signs associated with Parkinson's disease, and is a preferred target for many patients, particularly for those with troublesome medication induced dyskinesia," said Jerrold Vitek, M.D., Ph.D., head of the Neurology Department, director of the Neuromodulation Research Program, and center director of the University of Minnesota Udall Center of Excellence for Parkinson's Research. "This approval expands the options for patients to tailor treatment to their unique needs, with the added benefits of being able to target precise areas and utilizing a patient-friendly iOS device."
The system’s directional leads provide directed stimulation to areas of the brain to optimize patient outcomes and limit side effects. The platform can also upgrade the approved capabilities and new therapy features of the Infinity DBS system through simple, over-the-air updates.
Abbott continues to advance its Infinity DBS system with a focus on personalized and patient-centric innovations for people with movement disorders, supported by data from the PROGRESS study—the largest DBS study for directional lead use in Parkinson's disease.
"Abbott's PROGRESS study has led the way in establishing the value of directional DBS systems for targeted areas of the brain," noted Binith Cheeran, M.D., director of medical affairs, deep brain stimulation, a part of Abbott's neuromodulation business. "The approval of Abbott's Infinity DBS system with targeted stimulation is a significant advancement for people living with Parkinson's disease and their care teams. The ability to optimize the programming for each individual opens the door for a new standard of care for DBS therapy."
Data indicates patients gravitate toward intuitive, low-maintenance medical devices, such as the Infinity DBS system's dual channel recharge-free device.1 Abbott's Infinity DBS system also has CE mark and is available in nearly 30 countries.
Reference
1 Khaleeq T., Hasegawa H., Samuel M., Ashkan K. 2018. Fixed-Life or Rechargeable Battery for Deep Brain Stimulation: Which Do Patients Prefer? Neuromodulation 2018; E-pub ahead of print. DOI:10.1111/ner.12810
According to Abbott, its Infinity DBS is now the only directional DBS system approved for all major targets used to treat movement disorders, Parkinson's disease, and Essential Tremor: the subthalamic nucleus (STN), ventral intermediate nucleus (VIM), and GPi. The DBS system operates in these indications on an iOS software platform with Bluetooth. Clinicians can streamline the programming process with an iPad mini device using Abbott's Informity Programming feature to achieve optimal outcomes with directional leads. Patients can discreetly manage symptoms with the system’s iPod touch controller.
"The internal segment of the global pallidus, or GPi, is a well-established valuable DBS target for the management of the motor signs associated with Parkinson's disease, and is a preferred target for many patients, particularly for those with troublesome medication induced dyskinesia," said Jerrold Vitek, M.D., Ph.D., head of the Neurology Department, director of the Neuromodulation Research Program, and center director of the University of Minnesota Udall Center of Excellence for Parkinson's Research. "This approval expands the options for patients to tailor treatment to their unique needs, with the added benefits of being able to target precise areas and utilizing a patient-friendly iOS device."
The system’s directional leads provide directed stimulation to areas of the brain to optimize patient outcomes and limit side effects. The platform can also upgrade the approved capabilities and new therapy features of the Infinity DBS system through simple, over-the-air updates.
Abbott continues to advance its Infinity DBS system with a focus on personalized and patient-centric innovations for people with movement disorders, supported by data from the PROGRESS study—the largest DBS study for directional lead use in Parkinson's disease.
"Abbott's PROGRESS study has led the way in establishing the value of directional DBS systems for targeted areas of the brain," noted Binith Cheeran, M.D., director of medical affairs, deep brain stimulation, a part of Abbott's neuromodulation business. "The approval of Abbott's Infinity DBS system with targeted stimulation is a significant advancement for people living with Parkinson's disease and their care teams. The ability to optimize the programming for each individual opens the door for a new standard of care for DBS therapy."
Data indicates patients gravitate toward intuitive, low-maintenance medical devices, such as the Infinity DBS system's dual channel recharge-free device.1 Abbott's Infinity DBS system also has CE mark and is available in nearly 30 countries.
Reference
1 Khaleeq T., Hasegawa H., Samuel M., Ashkan K. 2018. Fixed-Life or Rechargeable Battery for Deep Brain Stimulation: Which Do Patients Prefer? Neuromodulation 2018; E-pub ahead of print. DOI:10.1111/ner.12810