Business Wire12.18.19
Penumbra, Inc., a global healthcare company focused on innovative therapies, announced that the EXTRACT-PE trial successfully met the primary endpoints, demonstrating the safety and efficacy of the Indigo Aspiration System for aspiration mechanical thrombectomy in patients with acute pulmonary embolism (PE).
“The positive results of the EXTRACT-PE trial, the first prospective safety and efficacy study of the Indigo Aspiration System, are a step forward in building needed clinical evidence around catheter-based embolectomy in patients with acute pulmonary embolism,” said Akhilesh Sista, M.D., FSIR, FAHA, EXTRACT-PE national principal investigator, chief of Vascular and Interventional Radiology at NYU-Langone School of Medicine, and vice chair of the recently published Interventional Therapies for Acute Pulmonary Embolism guidelines from the American Heart Association. “New AHA guidelines state that catheter-based embolectomy may have an important role in severe PE, but that more study is needed. EXTRACT-PE is an important first step in determining how and when we should employ promising and innovative catheter-based technologies such as the Indigo aspiration device.”
EXTRACT-PE was a prospective, single-arm, multicenter trial conducted under an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration (FDA) that evaluated the safety and efficacy of the Indigo Aspiration System in the treatment of acute pulmonary embolism. The study enrolled 119 participants across 22 U.S. study centers. The primary efficacy endpoint was met with a significant mean reduction in right ventricular (RV)/left ventricular (LV) ratio of 0.43, corresponding to a 27.3 percent reduction, at 48 hours after intervention. The primary safety endpoint was reached with a low major adverse event composite rate of 1.7 percent within 48 hours. The major adverse event composite included device-related death, major bleeding, and device-related serious adverse events as measured by the clinical events committee. Additional data reported included median time of Indigo System usage (37 minutes) and median ICU stay (one day). No thrombolytic drugs were used in 98.3 percent of patients.
“From a pulmonologist’s perspective, the results of the EXTRACT-PE trial are another important dataset that help broaden the mechanical treatment options for patients with pulmonary embolism,” said Victor F. Tapson, M.D., pulmonary critical care specialist at Cedars-Sinai Medical Center in Los Angeles and EXTRACT-PE steering committee member. “The safety profile of the Indigo System and the low rate of bleeding events, combined with the significant reduction in RV/LV ratio seen in the EXTRACT-PE trial, can positively impact the emerging field of acute pulmonary embolism intervention.”
“The Indigo Aspiration System has demonstrated tremendous potential to address patients with clot in other parts of the body, and today’s data show its specific potential in the pulmonary arteries,” said Adam Elsesser, chairman and CEO of Penumbra. “Pulmonary embolism is a deadly disease where we see an unmet clinical need and patient opportunity, and we commend the dedication of physicians who are focused on addressing this area. We are excited about the excellent results of this trial and look forward to working together with physicians in the field to bring better treatment options to patients.”
Pulmonary embolism (PE) is a condition that occurs when blood clots, typically traveling from the veins in the legs, get caught in the arteries of the lungs. Blood clots in the lungs can block the lungs from absorbing oxygen, causing strain on the heart and other organs. According to the American Heart Association, PE affects roughly 300,000 Americans per year.1
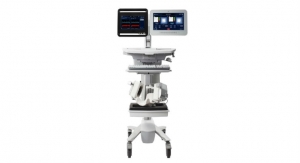
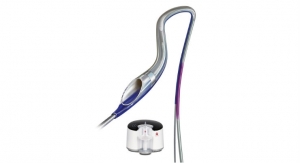
In 2014, Penumbra introduced the Indigo System, a continuous aspiration mechanical thrombectomy system designed to remove clot from arteries and veins in the peripheral vasculature. The Indigo System utilizes the Penumbra ENGINE aspiration source to deliver nearly pure, continuous vacuum suction to the Indigo System Aspiration Catheters to address thrombus in vessels of various sizes.
Penumbra Inc., headquartered in Alameda, Calif., is a global healthcare company that designs, develops, manufactures, and markets products and has a broad portfolio that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and vascular. Penumbra sells its products to hospitals primarily through its direct sales organization in the United States, most of Europe, Canada and Australia, and through distributors in select international markets.
Reference
1 Benjamin, E.J. et al. on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139:e56–e528.
“The positive results of the EXTRACT-PE trial, the first prospective safety and efficacy study of the Indigo Aspiration System, are a step forward in building needed clinical evidence around catheter-based embolectomy in patients with acute pulmonary embolism,” said Akhilesh Sista, M.D., FSIR, FAHA, EXTRACT-PE national principal investigator, chief of Vascular and Interventional Radiology at NYU-Langone School of Medicine, and vice chair of the recently published Interventional Therapies for Acute Pulmonary Embolism guidelines from the American Heart Association. “New AHA guidelines state that catheter-based embolectomy may have an important role in severe PE, but that more study is needed. EXTRACT-PE is an important first step in determining how and when we should employ promising and innovative catheter-based technologies such as the Indigo aspiration device.”
EXTRACT-PE was a prospective, single-arm, multicenter trial conducted under an Investigational Device Exemption (IDE) from the U.S. Food and Drug Administration (FDA) that evaluated the safety and efficacy of the Indigo Aspiration System in the treatment of acute pulmonary embolism. The study enrolled 119 participants across 22 U.S. study centers. The primary efficacy endpoint was met with a significant mean reduction in right ventricular (RV)/left ventricular (LV) ratio of 0.43, corresponding to a 27.3 percent reduction, at 48 hours after intervention. The primary safety endpoint was reached with a low major adverse event composite rate of 1.7 percent within 48 hours. The major adverse event composite included device-related death, major bleeding, and device-related serious adverse events as measured by the clinical events committee. Additional data reported included median time of Indigo System usage (37 minutes) and median ICU stay (one day). No thrombolytic drugs were used in 98.3 percent of patients.
“From a pulmonologist’s perspective, the results of the EXTRACT-PE trial are another important dataset that help broaden the mechanical treatment options for patients with pulmonary embolism,” said Victor F. Tapson, M.D., pulmonary critical care specialist at Cedars-Sinai Medical Center in Los Angeles and EXTRACT-PE steering committee member. “The safety profile of the Indigo System and the low rate of bleeding events, combined with the significant reduction in RV/LV ratio seen in the EXTRACT-PE trial, can positively impact the emerging field of acute pulmonary embolism intervention.”
“The Indigo Aspiration System has demonstrated tremendous potential to address patients with clot in other parts of the body, and today’s data show its specific potential in the pulmonary arteries,” said Adam Elsesser, chairman and CEO of Penumbra. “Pulmonary embolism is a deadly disease where we see an unmet clinical need and patient opportunity, and we commend the dedication of physicians who are focused on addressing this area. We are excited about the excellent results of this trial and look forward to working together with physicians in the field to bring better treatment options to patients.”
Pulmonary embolism (PE) is a condition that occurs when blood clots, typically traveling from the veins in the legs, get caught in the arteries of the lungs. Blood clots in the lungs can block the lungs from absorbing oxygen, causing strain on the heart and other organs. According to the American Heart Association, PE affects roughly 300,000 Americans per year.1
In 2014, Penumbra introduced the Indigo System, a continuous aspiration mechanical thrombectomy system designed to remove clot from arteries and veins in the peripheral vasculature. The Indigo System utilizes the Penumbra ENGINE aspiration source to deliver nearly pure, continuous vacuum suction to the Indigo System Aspiration Catheters to address thrombus in vessels of various sizes.
Penumbra Inc., headquartered in Alameda, Calif., is a global healthcare company that designs, develops, manufactures, and markets products and has a broad portfolio that addresses challenging medical conditions and significant clinical needs across two major markets, neuro and vascular. Penumbra sells its products to hospitals primarily through its direct sales organization in the United States, most of Europe, Canada and Australia, and through distributors in select international markets.
Reference
1 Benjamin, E.J. et al. on behalf of the American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. 2019;139:e56–e528.