Business Wire11.12.19
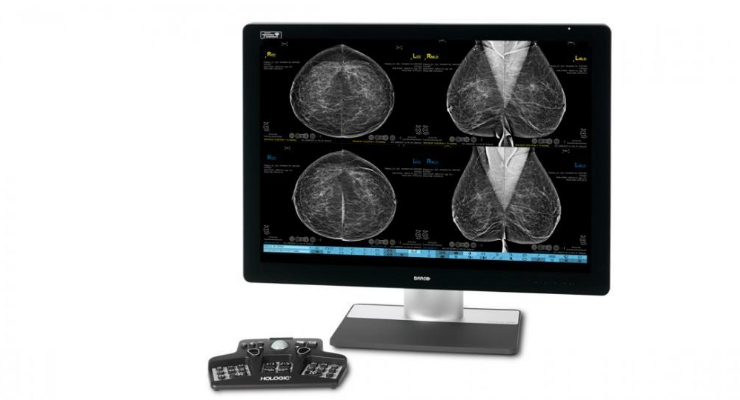
The FDA has approved Hologic Inc.’s 3DQuorum Imaging Technology, powered by Genius AI. 3DQuorum technology works in tandem with Hologic’s Clarity HD high resolution imaging technology to reduce tomosynthesis image volume for radiologists by 66 percent.2
In 2009, Hologic introduced the Genius 3D mammography exam, the only mammogram clinically proven and FDA approved as superior for all women, including those with dense breasts, compared to 2D mammography alone.3,4,5,6 Today, there are nearly 7,000 Hologic 3D Mammography systems in use in the U.S. alone, and tomosynthesis is now the standard of care for annual breast cancer screening. These advancements serve as the foundation for Genius AI technology, Hologic’s new core artificial intelligence platform designed to provide advanced cancer detection, operational efficiency and clinical decision support across the breast cancer care continuum.
3DQuorum technology uses Genius AI-powered analytics to uniquely reconstruct high-resolution 3D data to produce 6 mm “SmartSlices.” These analytics identify clinically relevant regions of interest and preserve important features during reconstruction of the SmartSlices.1,2 SmartSlices expedite read time by reducing the number of images for radiologists to review, without compromising image quality, sensitivity or accuracy.1,2 With 3DQuorum technology, the number of 3D images to review is reduced by two-thirds, saving an average of one hour per eight hours of daily image interpretation time.1,2,7
“Superior breast cancer detection begins with our superior imaging, and only Hologic offers a comprehensive breast health ecosystem that improves outcomes, reduces costs, and increases patient and clinician satisfaction,” said Pete Valenti, Hologic’s division president, Breast and Skeletal Health Solutions. “3DQuorum technology is the first of many products to come powered by our new Genius AI technology platform, which will further revolutionize cancer detection in the coming years.”
3DQuorum technology is available as part of the new Hologic Clarity HD Plus technology package for use with existing and future Hologic 3D mammography systems. More information can be found here.
References:
1 Tech File: TFL-00059
2 Report: CSR-00116
3 Results from Friedewald, SM, et al. "Breast cancer screening using tomosynthesis in combination with digital mammography." JAMA 311.24 (2014): 2499-2507; a multi-site (13), non-randomized, historical control study of 454,000 screening mammograms investigating the initial impact the introduction of the Hologic Selenia Dimensions on screening outcomes. Individual results may vary. The study found an average 41% increase and that 1.2 (95% CI: 0.8-1.6) additional invasive breast cancers per 1000 screening exams were found in women receiving combined 2D FFDM and 3D mammograms acquired with the Hologic 3D Mammography System versus women receiving 2D FFDM mammograms only.
4 Friedewald SM, Rafferty EA, Rose SL, Durand MA, Plecha DM, Greenberg JS, Hayes MK, Copit DS, Carlson KL, Cink TM, Carke LD, Greer LN, Miller DP, Conant EF, Breast Cancer Screening Using Tomosynthesis in Combination with Digital Mammography, JAMA June 25, 2014.
5 Bernardi D, Macaskill P, Pellegrini M, et al. Breast cancer screening with tomosynthesis (3D mammography) with acquired or synthetic 2D mammography compared with 2D mammography alone (STORM-2): a population-based prospective study. Lancet Oncol. 2016 Aug;17(8):1105-13.
6 FDA submissions P080003, P080003/S001, P080003/S004, P080003/S005
7 Physician Labeling: MAN-06153
In 2009, Hologic introduced the Genius 3D mammography exam, the only mammogram clinically proven and FDA approved as superior for all women, including those with dense breasts, compared to 2D mammography alone.3,4,5,6 Today, there are nearly 7,000 Hologic 3D Mammography systems in use in the U.S. alone, and tomosynthesis is now the standard of care for annual breast cancer screening. These advancements serve as the foundation for Genius AI technology, Hologic’s new core artificial intelligence platform designed to provide advanced cancer detection, operational efficiency and clinical decision support across the breast cancer care continuum.
3DQuorum technology uses Genius AI-powered analytics to uniquely reconstruct high-resolution 3D data to produce 6 mm “SmartSlices.” These analytics identify clinically relevant regions of interest and preserve important features during reconstruction of the SmartSlices.1,2 SmartSlices expedite read time by reducing the number of images for radiologists to review, without compromising image quality, sensitivity or accuracy.1,2 With 3DQuorum technology, the number of 3D images to review is reduced by two-thirds, saving an average of one hour per eight hours of daily image interpretation time.1,2,7
“Superior breast cancer detection begins with our superior imaging, and only Hologic offers a comprehensive breast health ecosystem that improves outcomes, reduces costs, and increases patient and clinician satisfaction,” said Pete Valenti, Hologic’s division president, Breast and Skeletal Health Solutions. “3DQuorum technology is the first of many products to come powered by our new Genius AI technology platform, which will further revolutionize cancer detection in the coming years.”
3DQuorum technology is available as part of the new Hologic Clarity HD Plus technology package for use with existing and future Hologic 3D mammography systems. More information can be found here.
References:
1 Tech File: TFL-00059
2 Report: CSR-00116
3 Results from Friedewald, SM, et al. "Breast cancer screening using tomosynthesis in combination with digital mammography." JAMA 311.24 (2014): 2499-2507; a multi-site (13), non-randomized, historical control study of 454,000 screening mammograms investigating the initial impact the introduction of the Hologic Selenia Dimensions on screening outcomes. Individual results may vary. The study found an average 41% increase and that 1.2 (95% CI: 0.8-1.6) additional invasive breast cancers per 1000 screening exams were found in women receiving combined 2D FFDM and 3D mammograms acquired with the Hologic 3D Mammography System versus women receiving 2D FFDM mammograms only.
4 Friedewald SM, Rafferty EA, Rose SL, Durand MA, Plecha DM, Greenberg JS, Hayes MK, Copit DS, Carlson KL, Cink TM, Carke LD, Greer LN, Miller DP, Conant EF, Breast Cancer Screening Using Tomosynthesis in Combination with Digital Mammography, JAMA June 25, 2014.
5 Bernardi D, Macaskill P, Pellegrini M, et al. Breast cancer screening with tomosynthesis (3D mammography) with acquired or synthetic 2D mammography compared with 2D mammography alone (STORM-2): a population-based prospective study. Lancet Oncol. 2016 Aug;17(8):1105-13.
6 FDA submissions P080003, P080003/S001, P080003/S004, P080003/S005
7 Physician Labeling: MAN-06153