Globe Newswire07.05.19
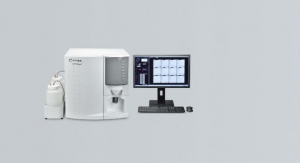
Cytek Biosciences Inc., a provider of advanced flow cytometry instrumentation, announced that its Quality Management System (QMS) has achieved ISO 9001:2015 certification. The certification applies to Cytek’s headquarters and manufacturing operations in Fremont, Calif., for the design, build, and support of flow cytometers and accessories.
Established by the International Organization for Standardization, ISO 9001:2015 is recognized as the worldwide standard for quality management systems. The certification body provides requirements, guidance and tools for companies and organizations that want to ensure that their products and services consistently meet customers’ requirements, and that quality is consistently improved. The ISO 9001:2015 certification provides a quality control process for Cytek to ensure high-quality operations and lays out a trackable, metrics-based roadmap of quality improvements.
“This certification is a direct reflection of our unwavering commitment to quality and customer service,” noted Dr. Wenbin Jiang, CEO of Cytek Biosciences. “By implementing processes in line with the standards of ISO 9001:2015, we are showing the world that we are dedicated to delivering the best quality solutions possible.”
Cytek’s ISO 9001:2015 certification follows extensive analysis of its QMS by TÜV Rheinland, an independent certification body. As its mission to make massively multiparametric flow cytometry accessible to a larger number of researchers marches on, Cytek will continue to pursue and obtain certifications for increasingly stringent standards and regulations.
Cytek Biosciences Inc. is a flow cytometry solution provider that develops compact instruments with high multiplexing capability and a range of services to support scientists and clinicians. Cytek has branch offices and distribution channels across the globe.
Established by the International Organization for Standardization, ISO 9001:2015 is recognized as the worldwide standard for quality management systems. The certification body provides requirements, guidance and tools for companies and organizations that want to ensure that their products and services consistently meet customers’ requirements, and that quality is consistently improved. The ISO 9001:2015 certification provides a quality control process for Cytek to ensure high-quality operations and lays out a trackable, metrics-based roadmap of quality improvements.
“This certification is a direct reflection of our unwavering commitment to quality and customer service,” noted Dr. Wenbin Jiang, CEO of Cytek Biosciences. “By implementing processes in line with the standards of ISO 9001:2015, we are showing the world that we are dedicated to delivering the best quality solutions possible.”
Cytek’s ISO 9001:2015 certification follows extensive analysis of its QMS by TÜV Rheinland, an independent certification body. As its mission to make massively multiparametric flow cytometry accessible to a larger number of researchers marches on, Cytek will continue to pursue and obtain certifications for increasingly stringent standards and regulations.
Cytek Biosciences Inc. is a flow cytometry solution provider that develops compact instruments with high multiplexing capability and a range of services to support scientists and clinicians. Cytek has branch offices and distribution channels across the globe.