Business Wire04.18.19
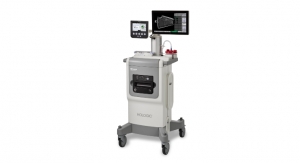
Hologic Inc. announced that the ThinPrep Genesis processor for cytology slide and molecular test preparation has received a CE IVD mark in Europe. The system features increased automation capabilities, together with ergonomic and chain of custody benefits, compared to older instruments. Most importantly, the instrument will prepare slides for cytology as well as aliquot samples for molecular testing, reducing manual sample handling.
The Genesis processor is the latest extension of the ThinPrep portfolio, which revolutionized and increased the accuracy of cervical cancer screening with the first liquid-based Pap test in 1996. Hologic launched an updated ThinPrep 2000 processor in 2017 and received FDA approval in 2018 for the ThinPrep Integrated Imager, which guides cytotechnologists to areas showing potential abnormal cells. Studies have shown that automated, assisted imaging can increase the sensitivity of detecting suspicious cells compared to manual slide review.1-4
“The launch of the ThinPrep Genesis processor reflects our ongoing commitment to helping our lab customers deliver accurate and reliable results to clinicians and patients, while maximizing their operational efficiency,” said Jan Verstreken, Hologic’s Regional President, EMEA and Canada. “Over the last 30 years, mortality from cervical cancer has fallen significantly in the developed world, largely due to screening programs that identify abnormalities before they become cervical cancer.”
The ThinPrep Genesis processor provides the same trusted reliability as the ThinPrep 2000 system for cytology slide processing, with added features such as automated aliquoting for molecular testing. The processor also provides automated barcoding of samples, ensuring accurate sample tracking as well as reducing manual steps. The increased automation is expected to deliver ergonomic benefits by reducing repetitive strain.
References
1 Dziura B, et al. Performance of an imaging system vs. manual screening in the detection of squamous intra epithelial lesions of the uterine cervix. Acta Cytol. 2006;50(3):309-11.
2 Lozano R. Comparison of computer-assisted and manual screening of cervical cytology. Gynecol Oncol. 2007;104(1):134-8.
3 Miller FS, et al. Implementation of the ThinPrep imaging system in a high-volume metropolitan laboratory. Diagn Cytopathol. 2007;35(4):213-7.4.
4 Klug SJ, et al. A randomized trial comparing conventional cytology to liquid-based cytology and computer assistance. Int J Cancer. 2012;132(12):2849-57. doi:10.1002/ijc.27955.
The Genesis processor is the latest extension of the ThinPrep portfolio, which revolutionized and increased the accuracy of cervical cancer screening with the first liquid-based Pap test in 1996. Hologic launched an updated ThinPrep 2000 processor in 2017 and received FDA approval in 2018 for the ThinPrep Integrated Imager, which guides cytotechnologists to areas showing potential abnormal cells. Studies have shown that automated, assisted imaging can increase the sensitivity of detecting suspicious cells compared to manual slide review.1-4
“The launch of the ThinPrep Genesis processor reflects our ongoing commitment to helping our lab customers deliver accurate and reliable results to clinicians and patients, while maximizing their operational efficiency,” said Jan Verstreken, Hologic’s Regional President, EMEA and Canada. “Over the last 30 years, mortality from cervical cancer has fallen significantly in the developed world, largely due to screening programs that identify abnormalities before they become cervical cancer.”
The ThinPrep Genesis processor provides the same trusted reliability as the ThinPrep 2000 system for cytology slide processing, with added features such as automated aliquoting for molecular testing. The processor also provides automated barcoding of samples, ensuring accurate sample tracking as well as reducing manual steps. The increased automation is expected to deliver ergonomic benefits by reducing repetitive strain.
References
1 Dziura B, et al. Performance of an imaging system vs. manual screening in the detection of squamous intra epithelial lesions of the uterine cervix. Acta Cytol. 2006;50(3):309-11.
2 Lozano R. Comparison of computer-assisted and manual screening of cervical cytology. Gynecol Oncol. 2007;104(1):134-8.
3 Miller FS, et al. Implementation of the ThinPrep imaging system in a high-volume metropolitan laboratory. Diagn Cytopathol. 2007;35(4):213-7.4.
4 Klug SJ, et al. A randomized trial comparing conventional cytology to liquid-based cytology and computer assistance. Int J Cancer. 2012;132(12):2849-57. doi:10.1002/ijc.27955.