Abbott Laboratories02.28.19
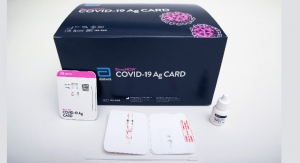
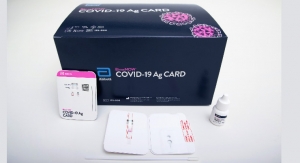
Abbott (announced the availability1 of its latest rapid influenza diagnostic test (RIDT), BinaxNOW Influenza A & B Card 2. The reformulated test has been granted waived status under the Clinical Laboratory Improvements Amendments (CLIA) by the U.S. Food and Drug Administration (FDA) for use with Abbott's DIGIVAL diagnostic reader (formerly Alere Reader) for the rapid detection of influenza virus.
The DIGIVAL reader accurately reads and interprets BinaxNOW Influenza A & B Card 2 tests in seconds, delivering automated, objective results in a broad range of healthcare settings. The combined platform is available for use in hospital laboratories, emergency rooms, physician offices, walk-in clinics, and urgent care centers throughout the U.S.
"This flu season, healthcare providers need objective tests that can provide fast, accurate results to determine the best course of care for patients," said Sharon Bracken, senior vice president, Rapid Diagnostics, Abbott. "The BinaxNOW Influenza A & B Card 2 adds to our strong portfolio of rapid flu tests by providing rapid and easy diagnosis, while DIGIVAL enhances healthcare providers' confidence in results by reducing user subjectivity."
"The BinaxNOW Influenza A & B Card 2 and DIGIVAL platform take the guesswork out of interpreting visually-read tests, empowering our staff to give an accurate influenza diagnosis and put patients on the path to recovery," said Marissa Ciminera, Strategic Sourcing and Procurement Manager, ProHealth. "We've installed DIGIVAL across our practices as it offers significant improvement over the subjective, visually-read tests we have historically used."
About BinaxNOW Influenza A & B Card 2 and Digival (Formerly Alere Reader)
The BinaxNOW Influenza A & B Card 2 test provides an advancement in lateral-flow testing technology. The card can be easily used by healthcare staff in physician offices and hospitals. The card is placed in the camera-based DIGIVAL diagnostic reader for result interpretation, which removes subjectivity and provides consistency in reading. In addition, the instrument helps with quality control management and can be connected to the hospital information system. As a Class II assay, the BinaxNOW Influenza A & B Card 2 complies with the FDA's new reclassification requirements for RIDTs. Abbott will offer assays for use with both existing Alere Reader instruments and new DIGIVAL instruments.
In addition to the BinaxNOW Influenza A & B Card 2, Abbott's rapid influenza test portfolio includes ID NOW, the first CLIA-waived point-of-care molecular platform used for the qualitative detection of influenza A & B, Strep A and respiratory syncytial virus (RSV).
Reference
1 The BinaxNOW Influenza A&B 2 assay is cleared by the FDA in United States, CE marked for sale in countries accepting CE mark with registrations pending for other markets outside U.S.
The DIGIVAL reader accurately reads and interprets BinaxNOW Influenza A & B Card 2 tests in seconds, delivering automated, objective results in a broad range of healthcare settings. The combined platform is available for use in hospital laboratories, emergency rooms, physician offices, walk-in clinics, and urgent care centers throughout the U.S.
"This flu season, healthcare providers need objective tests that can provide fast, accurate results to determine the best course of care for patients," said Sharon Bracken, senior vice president, Rapid Diagnostics, Abbott. "The BinaxNOW Influenza A & B Card 2 adds to our strong portfolio of rapid flu tests by providing rapid and easy diagnosis, while DIGIVAL enhances healthcare providers' confidence in results by reducing user subjectivity."
"The BinaxNOW Influenza A & B Card 2 and DIGIVAL platform take the guesswork out of interpreting visually-read tests, empowering our staff to give an accurate influenza diagnosis and put patients on the path to recovery," said Marissa Ciminera, Strategic Sourcing and Procurement Manager, ProHealth. "We've installed DIGIVAL across our practices as it offers significant improvement over the subjective, visually-read tests we have historically used."
About BinaxNOW Influenza A & B Card 2 and Digival (Formerly Alere Reader)
The BinaxNOW Influenza A & B Card 2 test provides an advancement in lateral-flow testing technology. The card can be easily used by healthcare staff in physician offices and hospitals. The card is placed in the camera-based DIGIVAL diagnostic reader for result interpretation, which removes subjectivity and provides consistency in reading. In addition, the instrument helps with quality control management and can be connected to the hospital information system. As a Class II assay, the BinaxNOW Influenza A & B Card 2 complies with the FDA's new reclassification requirements for RIDTs. Abbott will offer assays for use with both existing Alere Reader instruments and new DIGIVAL instruments.
In addition to the BinaxNOW Influenza A & B Card 2, Abbott's rapid influenza test portfolio includes ID NOW, the first CLIA-waived point-of-care molecular platform used for the qualitative detection of influenza A & B, Strep A and respiratory syncytial virus (RSV).
Reference
1 The BinaxNOW Influenza A&B 2 assay is cleared by the FDA in United States, CE marked for sale in countries accepting CE mark with registrations pending for other markets outside U.S.