Globe Newswire02.05.19
IRADIMED Corporation, developer of magnetic resonance imaging (MRI) medical devices, has received U.S. Food and Drug Administration 510(k) clearance of its 3883 MRI-compatible invasive blood pressure module for use with its 3880 MRI compatible patient vital signs monitoring system.
“This newly cleared device represents a feature extension to the 3880 MRI-compatible patient monitoring system, which we launched in the U.S. just over a year ago. With the invasive blood pressure module, we now offer advanced monitoring capabilities to our customers,” said Roger Susi, president and CEO of the company.
The 3883 MRI-compatible invasive blood pressure module is available as an option to the 3880 MRI-compatible patient vital signs monitor.
“Customers can now provide a more comprehensive suite of vital signs monitoring while transporting patients from hospital critical care departments to the MRI scanner room and back to critical care while maintaining continuous vital signs monitoring,” said Susi.
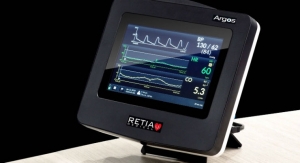
IRADIMED designed the 3880 MRI-compatible patient vital signs monitoring system using non-magnetic components and other special features enabling the safe and accurate monitoring of a patient’s vital signs during various MRI procedures.
The IRADIMED 3880 MRI-compatible patient vital signs monitoring system has an easy-to-use design and allows for the effective communication of patient vital signs information to clinicians.
Additionally, the IRADIMED 3880 MRI-compatible vital signs monitoring system’s compact and lightweight design enables uninterrupted monitoring during transport, resulting in increased patient safety and decreasing the amount of time critically ill patients are away from critical care units.
Available features of the IRADIMED 3880 system include:
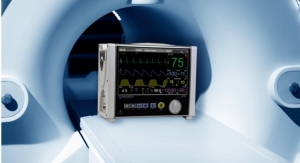
The IRADIMED 3880 system operates dependably in magnetic fields up to 30,000 gauss, which means it can operate virtually anywhere in the MRI scanner room.
IRADIMED Corporation develops MRI-compatible medical devices. The company is the only known provider of a non-magnetic intravenous infusion pump system that is specifically designed to be safe for use during MRI procedures. IRADIMED was the first to develop an infusion delivery system that largely eliminates many of the dangers and problems present during MRI procedures. Standard infusion pumps contain magnetic and electronic components which can create radio frequency interference and are dangerous to operate in the presence of the powerful magnet that drives an MRI system. The company's patented MRidium MRI-compatible IV infusion pump system has been designed with a non-magnetic ultrasonic motor, uniquely-designed non-ferrous parts and other special features to safely and predictably deliver anesthesia and other IV fluids during various MRI procedures. IRADIMED's pump solution provides a seamless approach that enables accurate, safe and dependable fluid delivery before, during and after an MRI scan, which is important to critically-ill patients who cannot be removed from their vital medications, and children and infants who must generally be sedated to remain immobile during an MRI scan.
“This newly cleared device represents a feature extension to the 3880 MRI-compatible patient monitoring system, which we launched in the U.S. just over a year ago. With the invasive blood pressure module, we now offer advanced monitoring capabilities to our customers,” said Roger Susi, president and CEO of the company.
The 3883 MRI-compatible invasive blood pressure module is available as an option to the 3880 MRI-compatible patient vital signs monitor.
“Customers can now provide a more comprehensive suite of vital signs monitoring while transporting patients from hospital critical care departments to the MRI scanner room and back to critical care while maintaining continuous vital signs monitoring,” said Susi.
IRADIMED designed the 3880 MRI-compatible patient vital signs monitoring system using non-magnetic components and other special features enabling the safe and accurate monitoring of a patient’s vital signs during various MRI procedures.
The IRADIMED 3880 MRI-compatible patient vital signs monitoring system has an easy-to-use design and allows for the effective communication of patient vital signs information to clinicians.
Additionally, the IRADIMED 3880 MRI-compatible vital signs monitoring system’s compact and lightweight design enables uninterrupted monitoring during transport, resulting in increased patient safety and decreasing the amount of time critically ill patients are away from critical care units.
Available features of the IRADIMED 3880 system include:
- Wireless ECG with dynamic gradient filtering;
- Wireless SpO2 using Masimo SET algorithms;
- Non-magnetic respiratory CO2;
- Non-invasive blood pressure;
- Invasive blood pressure;
- Patient temperature;
- Advanced Masimo multi-gas anesthetic agent unit featuring continuous Minimum Alveolar Concentration measurements.
The IRADIMED 3880 system operates dependably in magnetic fields up to 30,000 gauss, which means it can operate virtually anywhere in the MRI scanner room.
IRADIMED Corporation develops MRI-compatible medical devices. The company is the only known provider of a non-magnetic intravenous infusion pump system that is specifically designed to be safe for use during MRI procedures. IRADIMED was the first to develop an infusion delivery system that largely eliminates many of the dangers and problems present during MRI procedures. Standard infusion pumps contain magnetic and electronic components which can create radio frequency interference and are dangerous to operate in the presence of the powerful magnet that drives an MRI system. The company's patented MRidium MRI-compatible IV infusion pump system has been designed with a non-magnetic ultrasonic motor, uniquely-designed non-ferrous parts and other special features to safely and predictably deliver anesthesia and other IV fluids during various MRI procedures. IRADIMED's pump solution provides a seamless approach that enables accurate, safe and dependable fluid delivery before, during and after an MRI scan, which is important to critically-ill patients who cannot be removed from their vital medications, and children and infants who must generally be sedated to remain immobile during an MRI scan.