PR Newswire02.05.19
CSA Medical Inc. announced that Dr. John T. Maple, a gastroenterologist at the University of Oklahoma College of Medicine, completed the state's first two procedures in patients with esophageal disease using truFreeze Spray Cryotherapy. This advanced technology is the only U.S. Food and Drug Administration-cleared option to treat patients across the spectrum of esophageal diseases, including esophageal cancer, as well as the precursor condition, Barrett's esophagus.
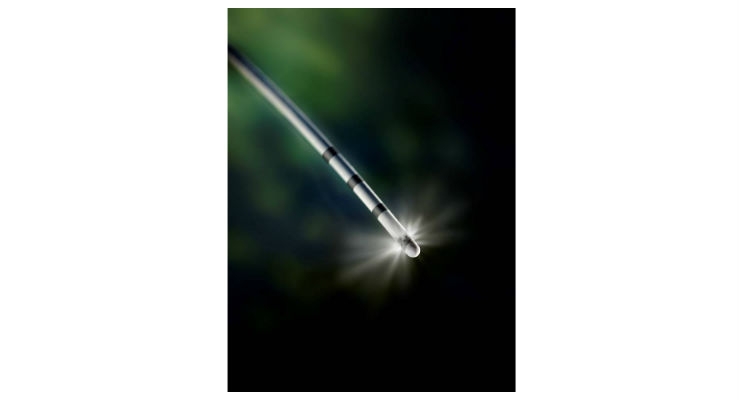
The truFreeze Spray Cryotherapy platform uses the extreme cold of liquid nitrogen (-196 degrees Celsius) to provide targeted, touchless ablation of diseased tissue, including cancer, while preserving underlying healthy tissue— in contrast to the scar tissue that forms after heat-based modalities such as radio-frequency ablation (RFA). In addition, truFreeze Spray Cryotherapy is unlike other warmer cryogens such as nitrous oxide (-88 degrees Celsius) which can't get tissue cold enough, fast enough to penetrate and ablate cancer.
Dr. Maple performed liquid nitrogen spray cryotherapy on two patients. One patient had persistent nodular (or bumpy) Barrett's esophagus, but an esophageal stricture related to a prior endoscopic mucosal resection (EMR) precluded further resection. The second patient had previously undergone chemotherapy and radiotherapy for esophageal adenocarcinoma and had a small amount of cancer remaining amenable to spray cryotherapy. Both patients experienced minimal discomfort during and after the procedures, and were treated on an outpatient basis with no complications.
"Ablation is one of several modalities that we use to treat dysplastic BE. Because it's a spray, the cryoablation is not dependent on the disease topography, and spray cryotherapy is associated with minimal pain, faster recovery, and less harm to healthy tissue," said Dr. Maple.
"Dr. Maple's recent spray cryotherapy procedures emphasize truFreeze's benefit to the gastroenterology community," said Wendelin Maners, CSA's president, truFreeze Business. "Because truFreeze is the only FDA-cleared esophageal ablation technology proven to effectively treat the entire esophageal disease spectrum, our hope is that other institutions follow suit and incorporate spray cryotherapy as their first ablative treatment for diseases of the GI tract."
CSA Medical Inc. develops and manufactures proprietary, interventional spray cryotherapy technology platforms harnessing the power of liquid nitrogen spray delivered by software driven devices with specialty catheters. Extremely cold (-196 degrees Celsius) liquid nitrogen spray flash freezes and destroys unwanted tissue while enabling a rejuvenative pattern of healing. CSA manufactures and distributes the truFreeze system in the United States, which is currently being utilized in over 100 hospitals and universities advancing therapy for patients affected by conditions such as Barrett's esophagus, esophageal cancer, and benign and malignant airway obstructions.
The truFreeze Spray Cryotherapy platform uses the extreme cold of liquid nitrogen (-196 degrees Celsius) to provide targeted, touchless ablation of diseased tissue, including cancer, while preserving underlying healthy tissue— in contrast to the scar tissue that forms after heat-based modalities such as radio-frequency ablation (RFA). In addition, truFreeze Spray Cryotherapy is unlike other warmer cryogens such as nitrous oxide (-88 degrees Celsius) which can't get tissue cold enough, fast enough to penetrate and ablate cancer.
Dr. Maple performed liquid nitrogen spray cryotherapy on two patients. One patient had persistent nodular (or bumpy) Barrett's esophagus, but an esophageal stricture related to a prior endoscopic mucosal resection (EMR) precluded further resection. The second patient had previously undergone chemotherapy and radiotherapy for esophageal adenocarcinoma and had a small amount of cancer remaining amenable to spray cryotherapy. Both patients experienced minimal discomfort during and after the procedures, and were treated on an outpatient basis with no complications.
"Ablation is one of several modalities that we use to treat dysplastic BE. Because it's a spray, the cryoablation is not dependent on the disease topography, and spray cryotherapy is associated with minimal pain, faster recovery, and less harm to healthy tissue," said Dr. Maple.
"Dr. Maple's recent spray cryotherapy procedures emphasize truFreeze's benefit to the gastroenterology community," said Wendelin Maners, CSA's president, truFreeze Business. "Because truFreeze is the only FDA-cleared esophageal ablation technology proven to effectively treat the entire esophageal disease spectrum, our hope is that other institutions follow suit and incorporate spray cryotherapy as their first ablative treatment for diseases of the GI tract."
CSA Medical Inc. develops and manufactures proprietary, interventional spray cryotherapy technology platforms harnessing the power of liquid nitrogen spray delivered by software driven devices with specialty catheters. Extremely cold (-196 degrees Celsius) liquid nitrogen spray flash freezes and destroys unwanted tissue while enabling a rejuvenative pattern of healing. CSA manufactures and distributes the truFreeze system in the United States, which is currently being utilized in over 100 hospitals and universities advancing therapy for patients affected by conditions such as Barrett's esophagus, esophageal cancer, and benign and malignant airway obstructions.