Globe Newswire01.16.19
BioSig Technologies Inc. has announced that the company formed a Clinical Advisory Board to complement existing scientific relationships with nationwide key opinion leaders built during the R&D phase of product development.
The goal of the newly formed board is to advise the company on a range of aspects related to commercialization of BioSig’s novel signal acquisition and processing technology, PURE EP System. Physicians from 17 leading medical centers in the United States gathered for the board’s first meeting on Nov. 30, 2018. The agenda included focused consultations on the human clinical data collection, commercial strategy for PURE EP System and potential clinical benefits of new technologies in the R&D pipeline.
“Our technology has been developed together with some of the most respected key opinion leaders in the electrophysiology industry. As we progress towards full commercial launch of our PURE EP System, it is essential to engage a wider group of talented physicians who can strengthen our product and launch plan. We are pleased that so many leading electrophysiologists agreed to dedicate their valuable time and provide input to help us better address unmet clinical needs in this very complex area of arrhythmia treatments,” commented Kenneth L. Londoner, chairman and CEO of BioSig Technologies Inc.
BioSig received U.S. Food and Drug Administration (FDA) 510(k) clearance for its first product, PURE EP System on Aug. 8, 2018. The company began trading on the Nasdaq Capital Markets on Sept. 21, 2018, under the ticker symbol BSGM.
BioSig Technologies is a medical technology company developing a proprietary biomedical signal processing platform designed to improve the electrophysiology (EP) marketplace. Los Angeles, Calif.-based BioSig Technologies is preparing to commercialize its PURE EP System. The technology has been developed to address an unmet need in a large and growing market.
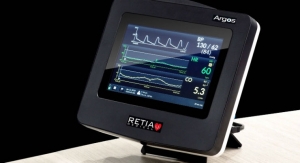
The company’s first product, PURE EP System, is a cardiac signal acquisition and display system which is engineered to assist electrophysiologists in clinical decision-making during procedures to diagnose and treat patients with abnormal heart rates and rhythms. BioSig’s main goal is to deliver technology to improve upon catheter ablation treatments for the prevalent and potentially deadly arrhythmias, atrial fibrillation and ventricular tachycardia. BioSig has partnered with Minnetronix on technology development.
The goal of the newly formed board is to advise the company on a range of aspects related to commercialization of BioSig’s novel signal acquisition and processing technology, PURE EP System. Physicians from 17 leading medical centers in the United States gathered for the board’s first meeting on Nov. 30, 2018. The agenda included focused consultations on the human clinical data collection, commercial strategy for PURE EP System and potential clinical benefits of new technologies in the R&D pipeline.
“Our technology has been developed together with some of the most respected key opinion leaders in the electrophysiology industry. As we progress towards full commercial launch of our PURE EP System, it is essential to engage a wider group of talented physicians who can strengthen our product and launch plan. We are pleased that so many leading electrophysiologists agreed to dedicate their valuable time and provide input to help us better address unmet clinical needs in this very complex area of arrhythmia treatments,” commented Kenneth L. Londoner, chairman and CEO of BioSig Technologies Inc.
BioSig received U.S. Food and Drug Administration (FDA) 510(k) clearance for its first product, PURE EP System on Aug. 8, 2018. The company began trading on the Nasdaq Capital Markets on Sept. 21, 2018, under the ticker symbol BSGM.
BioSig Technologies is a medical technology company developing a proprietary biomedical signal processing platform designed to improve the electrophysiology (EP) marketplace. Los Angeles, Calif.-based BioSig Technologies is preparing to commercialize its PURE EP System. The technology has been developed to address an unmet need in a large and growing market.
The company’s first product, PURE EP System, is a cardiac signal acquisition and display system which is engineered to assist electrophysiologists in clinical decision-making during procedures to diagnose and treat patients with abnormal heart rates and rhythms. BioSig’s main goal is to deliver technology to improve upon catheter ablation treatments for the prevalent and potentially deadly arrhythmias, atrial fibrillation and ventricular tachycardia. BioSig has partnered with Minnetronix on technology development.