Globe Newswire12.26.18
Physeon GmbH, a developer of medical technology products for venous access, has enrolled the first patient in its “VIVA” (Veinplicity for Improved Venous Access) Trial in the United States for the Veinplicity Device.
“We are very pleased to announce our first patient enrolled in the VIVA trial. This is the most important milestone for Veinplicity as we work towards what we believe will be a successful U.S. randomized clinical trial. We anticipate that the trial will move ahead in short order since this is not a complex trial and is straight forward in the metrics for success.” said Patrick Kullmann, CEO of Physeon.
The VIVA Trial design is similar to a recently conducted European Union randomized trial structure. It is randomized using standard tourniquet vs. Veinplicity + tourniquet in 246 patients. The primary end point is first stick success for superiority. Other key end points include number of attempted sticks to successful vein access, total procedure time, subject and clinician satisfaction scoring and change in the vein quality score from baseline to post-stimulation as well as health care economics.
Veinplicity is designed to alleviate one of the most common challenges in health care— difficult venous access (DVA). Up to 80 percent of hospitalized patients need a peripheral IV cannula for the infusion of fluids and medications. Although it’s the most common invasive procedure performed in health care, first attempt failure rates can be high, costly, cause needless damage to veins and lead to increased stress for patients and staff. It can also be an administrative and financial burden for both hospitals and outpatient centers.
Veinplicity is intended to address these challenges by significantly increasing vessel size, vessel wall rigidity and stability of targeted peripheral veins, making them easier to find and easier to cannulate for an IV at first attempt. It is the only technology available which has a positive and temporary physiological effect on target veins for the placement of IVs by making the targeted vein temporarily larger, resulting in making it an easier target to hit on a first attempt to cannulate.
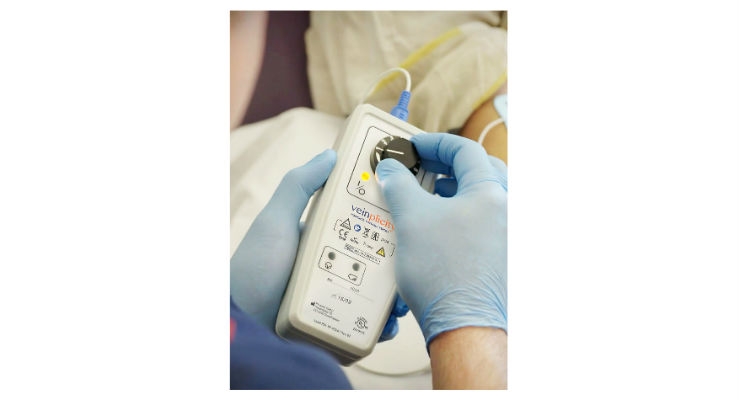
Veinplicity consists of a proprietary portable electronic stimulation device which passes a gentle electrical current between proprietary and dedicated electrodes placed on the palm of the hand and the bicep area of the patient.
In one of the company’s initial clinical studies, the dilatory effect of Veinplicity was compared to the dilatory effect of heat pack treatment, which has long been considered the standard of care when veins cannot be palpated. As well as dilating veins 38 percent more, Veinplicity’ s effect lasted twice as long as heat treatment, allowing clinicians crucial extra time to perform this important procedure.
The Veinplicity Device is CE Marked and is available in several countries in the European Union. It is not yet approved for use in the United States by the U.S. Food and Drug Administration.
Established in 2015 and located in Schaffhausen, Switzerland, Physeon is a boutique medical device company owned by Novintum Medical Technology GmbH. It was created to guide the development and commercialization of new medical innovations. The company embraces research and science to bring about innovative ideas and medical products that can advance the health and well-being of patients and simplify processes for healthcare professionals.
“We are very pleased to announce our first patient enrolled in the VIVA trial. This is the most important milestone for Veinplicity as we work towards what we believe will be a successful U.S. randomized clinical trial. We anticipate that the trial will move ahead in short order since this is not a complex trial and is straight forward in the metrics for success.” said Patrick Kullmann, CEO of Physeon.
The VIVA Trial design is similar to a recently conducted European Union randomized trial structure. It is randomized using standard tourniquet vs. Veinplicity + tourniquet in 246 patients. The primary end point is first stick success for superiority. Other key end points include number of attempted sticks to successful vein access, total procedure time, subject and clinician satisfaction scoring and change in the vein quality score from baseline to post-stimulation as well as health care economics.
Veinplicity is designed to alleviate one of the most common challenges in health care— difficult venous access (DVA). Up to 80 percent of hospitalized patients need a peripheral IV cannula for the infusion of fluids and medications. Although it’s the most common invasive procedure performed in health care, first attempt failure rates can be high, costly, cause needless damage to veins and lead to increased stress for patients and staff. It can also be an administrative and financial burden for both hospitals and outpatient centers.
Veinplicity is intended to address these challenges by significantly increasing vessel size, vessel wall rigidity and stability of targeted peripheral veins, making them easier to find and easier to cannulate for an IV at first attempt. It is the only technology available which has a positive and temporary physiological effect on target veins for the placement of IVs by making the targeted vein temporarily larger, resulting in making it an easier target to hit on a first attempt to cannulate.
Veinplicity consists of a proprietary portable electronic stimulation device which passes a gentle electrical current between proprietary and dedicated electrodes placed on the palm of the hand and the bicep area of the patient.
In one of the company’s initial clinical studies, the dilatory effect of Veinplicity was compared to the dilatory effect of heat pack treatment, which has long been considered the standard of care when veins cannot be palpated. As well as dilating veins 38 percent more, Veinplicity’ s effect lasted twice as long as heat treatment, allowing clinicians crucial extra time to perform this important procedure.
The Veinplicity Device is CE Marked and is available in several countries in the European Union. It is not yet approved for use in the United States by the U.S. Food and Drug Administration.
Established in 2015 and located in Schaffhausen, Switzerland, Physeon is a boutique medical device company owned by Novintum Medical Technology GmbH. It was created to guide the development and commercialization of new medical innovations. The company embraces research and science to bring about innovative ideas and medical products that can advance the health and well-being of patients and simplify processes for healthcare professionals.