Business Wire12.17.18
OcuMedic Inc., the developer of new, drug-eluting soft contact lenses and clear, corneal bandage technology, announced the European Patent Office has granted the patent Contact Drug Delivery System covering the company’s intellectual property for timed-released delivery of drugs directly to the eye through prescription, soft contact lenses.
The claims allowed by the patent cover extended-wear contacts that deliver a wide-variety of drugs for the treatment of conditions such as glaucoma and dry eye. The contacts also provide controlled release of anti-inflammatory, anti-biotic and pain-reducing drugs for post-operative care for procedures such as cataract surgery and LASIK.
“The European patent extends the company’s intellectual property coverage to the important European market valued at $16.7 billion annually, growing by 5.6 percent CAGR by 2023 according to Allied Market Research,” said Keith D. Ignotz, president and CEO of OcuMedic Inc. “This latest grant brings to 10 the number of patents in our portfolio, covering the two largest targeted markets— Europe and the United States. The technology places the delivery of ophthalmic drugs in the hands of the physician, relieving the patient of the therapeutic burden of having to remember to self-deliver drops while guaranteeing the drug is reaching the target tissue.”
“This additional patent is an important validation of our novel approach to delivering the best eye care possible to a large number of people suffering from poor vision or diseases of the eye,” said Mark E. Byrne, Ph.D., founding head and professor of Biomedical Engineering at Rowan University, inventor of the technology, and previously, a distinguished professor at Auburn University, and chief technical officer of OcuMedic Inc. “Our goal is to extend and enhance the gift of sight for as many as possible, and our new technology provides a better way to better eye health.”
Approximately 6 million Americans undergo some type of eye surgery every year—4 million cataract, 1 million post-corneal abrasion surgeries, and 800,000 Lasik procedures. More than 3 million people in Europe undergo cataract surgery, annually. All patients require post-operative care, which includes eye drops that can wash out quickly with only about 5 percent of the therapy reaching the target tissue. Eye drops can be hard to administer, and the low delivered concentration of medication tends to aggravate post-operative complications, costing $70 billion per year. Dry eye disease affects greater than 5 million people in the United States and approximately 40 million worldwide and is the most frequent complaint and reason to visit an eye care professional in the United States. Approximately 3 million Americans suffer from Glaucoma; worldwide, more than 60 million people are affected.
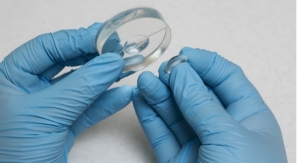
OcuMedic Inc. is the developer of the world’s first, drug-eluting therapeutic lens/clear corneal bandage for the delivery of drugs for the global ophthalmic market. Invented by Byrne, the therapeutic lens is a soft contact developed in the most widely used safe lens material that precisely controls the release of a “first line” anti-inflammatory drug that targets post-operative eye care to reduce healing time and costly complications. The device is a platform technology, with OcuMedic demonstrating controlled release of a number of therapeutics for the duration of wear, targeted at the large and growing surgery, dry eye, corneal abrasion, and glaucoma markets.
The claims allowed by the patent cover extended-wear contacts that deliver a wide-variety of drugs for the treatment of conditions such as glaucoma and dry eye. The contacts also provide controlled release of anti-inflammatory, anti-biotic and pain-reducing drugs for post-operative care for procedures such as cataract surgery and LASIK.
“The European patent extends the company’s intellectual property coverage to the important European market valued at $16.7 billion annually, growing by 5.6 percent CAGR by 2023 according to Allied Market Research,” said Keith D. Ignotz, president and CEO of OcuMedic Inc. “This latest grant brings to 10 the number of patents in our portfolio, covering the two largest targeted markets— Europe and the United States. The technology places the delivery of ophthalmic drugs in the hands of the physician, relieving the patient of the therapeutic burden of having to remember to self-deliver drops while guaranteeing the drug is reaching the target tissue.”
“This additional patent is an important validation of our novel approach to delivering the best eye care possible to a large number of people suffering from poor vision or diseases of the eye,” said Mark E. Byrne, Ph.D., founding head and professor of Biomedical Engineering at Rowan University, inventor of the technology, and previously, a distinguished professor at Auburn University, and chief technical officer of OcuMedic Inc. “Our goal is to extend and enhance the gift of sight for as many as possible, and our new technology provides a better way to better eye health.”
Approximately 6 million Americans undergo some type of eye surgery every year—4 million cataract, 1 million post-corneal abrasion surgeries, and 800,000 Lasik procedures. More than 3 million people in Europe undergo cataract surgery, annually. All patients require post-operative care, which includes eye drops that can wash out quickly with only about 5 percent of the therapy reaching the target tissue. Eye drops can be hard to administer, and the low delivered concentration of medication tends to aggravate post-operative complications, costing $70 billion per year. Dry eye disease affects greater than 5 million people in the United States and approximately 40 million worldwide and is the most frequent complaint and reason to visit an eye care professional in the United States. Approximately 3 million Americans suffer from Glaucoma; worldwide, more than 60 million people are affected.
OcuMedic Inc. is the developer of the world’s first, drug-eluting therapeutic lens/clear corneal bandage for the delivery of drugs for the global ophthalmic market. Invented by Byrne, the therapeutic lens is a soft contact developed in the most widely used safe lens material that precisely controls the release of a “first line” anti-inflammatory drug that targets post-operative eye care to reduce healing time and costly complications. The device is a platform technology, with OcuMedic demonstrating controlled release of a number of therapeutics for the duration of wear, targeted at the large and growing surgery, dry eye, corneal abrasion, and glaucoma markets.