Business Wire12.11.18
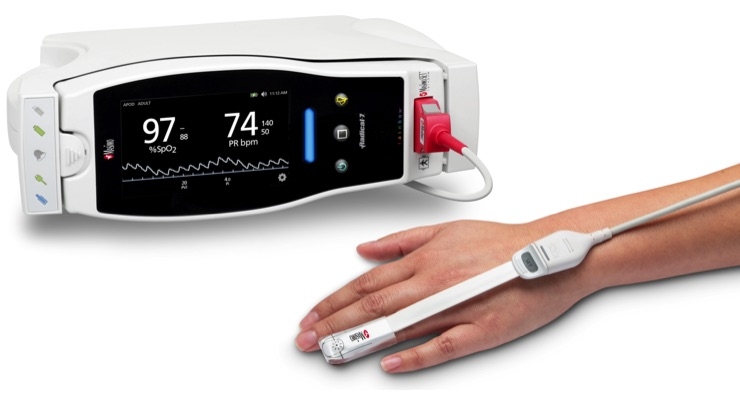
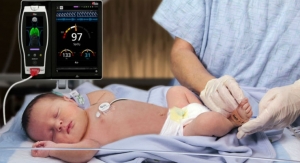
Masimo announced that RD SET sensors with Masimo Measure-through Motion and Low Perfusion SET pulse oximetry have received U.S. Food and Drug Administration (FDA) clearance with improved SpO2 accuracy specifications for all patients > 3 kg. RD SET single-patient-use sensors with the improved accuracy specifications are now available. The new RD SET sensors’ SpO2 accuracy specifications during patient motion have improved for adult, pediatric, and infant patients to 1.5 percent (at 1 SD), compared to previous accuracy specifications of 3 percent.
In addition to offering improved accuracy, RD SET sensors are designed to enhance patient comfort, optimize clinician workflows, and help hospitals meet green initiatives. The sensors are lightweight and have a flat, soft cable with smooth edges, so that they lie comfortably on a patient’s hand or foot. The sensors feature an intuitive sensor-to-cable connection. Their lightweight design results in up to 84 percent less waste, and their sleek, recyclable packaging reduces storage and shipping space.
The clinical benefits of Masimo’s revolutionary SET pulse oximetry have been demonstrated in a variety of clinical conditions. Studies have shown:
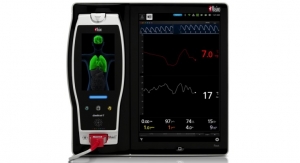
In addition to excellent accuracy and reliability, the SET platform with rainbow is also the only oximetry technology that also allows clinicians to measure physiological parameters such as total hemoglobin, carboxyhemoglobin, methemoglobin, and PVi.
Masimo is a global leader in noninvasive monitoring technologies. Our mission is to improve patient outcomes and reduce the cost of care. In 1995, the company debuted Masimo SET Measure-through Motion and Low Perfusion pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,1 improve CCHD screening in newborns,3 and, when used for continuous monitoring with Masimo Patient SafetyNet in post-surgical wards, reduce rapid response activations and costs.5-7 Masimo SET is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world,8 and is the primary pulse oximetry at nine of the top 10 hospitals listed in the 2018-19 U.S. News and World Report Best Hospitals Honor Roll.9 In 2005, Masimo introduced rainbow Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb), oxygen content (SpOC), carboxyhemoglobin (SpCO), methemoglobin (SpMet), Pleth Variability Index (PVi), and more recently, Oxygen Reserve Index (ORi), in addition to SpO2, pulse rate, and perfusion index (Pi). In 2014, Masimo introduced Root, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect (MOC-9) interface, enabling other companies to augment Root with new features and measurement capabilities. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7 wearable patient monitor, iSpO2 pulse oximeter for smartphones, and the MightySat fingertip pulse oximeter.
ORi has not received FDA 510(k) clearance and is not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
1. Castillo et al. Prevention of retinopathy of prematurity in preterm infants through changes in clinical practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
2. Zhao et al. Pulse oximetry with clinical assessment to screen for congenital heart disease in neonates in China: a prospective study. Lancet. 2014 Aug 30;384(9945):747-54.
3. de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;Jan 8;338.
4. Ewer A et al. Pulse Oximetry Screening for Congenital Heart Defects in Newborn Infants (Pulseox): A Test Accuracy Study. Lancet. 2011 Aug 27;378(9793):785-94.
5. Taenzer AH et al. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology. 2010:112(2):282-287.
6. Taenzer A et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
7. McGrath SP et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
8. Estimate: Masimo data on file.
9. http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.
In addition to offering improved accuracy, RD SET sensors are designed to enhance patient comfort, optimize clinician workflows, and help hospitals meet green initiatives. The sensors are lightweight and have a flat, soft cable with smooth edges, so that they lie comfortably on a patient’s hand or foot. The sensors feature an intuitive sensor-to-cable connection. Their lightweight design results in up to 84 percent less waste, and their sleek, recyclable packaging reduces storage and shipping space.
The clinical benefits of Masimo’s revolutionary SET pulse oximetry have been demonstrated in a variety of clinical conditions. Studies have shown:
- Masimo SET helped significantly reduce rates of severe retinopathy of prematurity (ROP) and the need for laser treatment.1
- Masimo SET-enabled pulse oximetry screening for critical congenital heart disease (CCHD), helping save many full-term newborns’ lives whose disease would otherwise have gone undiagnosed.2-4
- With Masimo SET on post-surgical wards, rescue calls and ICU transfers were reduced by 65 percent and 48 percent, respectively.5 Post-operative continuous surveillance monitoring, combined with Masimo SET, led to zero preventable deaths or brain damage due to opioids over five years.6
In addition to excellent accuracy and reliability, the SET platform with rainbow is also the only oximetry technology that also allows clinicians to measure physiological parameters such as total hemoglobin, carboxyhemoglobin, methemoglobin, and PVi.
Masimo is a global leader in noninvasive monitoring technologies. Our mission is to improve patient outcomes and reduce the cost of care. In 1995, the company debuted Masimo SET Measure-through Motion and Low Perfusion pulse oximetry, which has been shown in multiple studies to significantly reduce false alarms and accurately monitor for true alarms. Masimo SET has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,1 improve CCHD screening in newborns,3 and, when used for continuous monitoring with Masimo Patient SafetyNet in post-surgical wards, reduce rapid response activations and costs.5-7 Masimo SET is estimated to be used on more than 100 million patients in leading hospitals and other healthcare settings around the world,8 and is the primary pulse oximetry at nine of the top 10 hospitals listed in the 2018-19 U.S. News and World Report Best Hospitals Honor Roll.9 In 2005, Masimo introduced rainbow Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb), oxygen content (SpOC), carboxyhemoglobin (SpCO), methemoglobin (SpMet), Pleth Variability Index (PVi), and more recently, Oxygen Reserve Index (ORi), in addition to SpO2, pulse rate, and perfusion index (Pi). In 2014, Masimo introduced Root, an intuitive patient monitoring and connectivity platform with the Masimo Open Connect (MOC-9) interface, enabling other companies to augment Root with new features and measurement capabilities. Masimo is also taking an active leadership role in mHealth with products such as the Radius-7 wearable patient monitor, iSpO2 pulse oximeter for smartphones, and the MightySat fingertip pulse oximeter.
ORi has not received FDA 510(k) clearance and is not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
1. Castillo et al. Prevention of retinopathy of prematurity in preterm infants through changes in clinical practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
2. Zhao et al. Pulse oximetry with clinical assessment to screen for congenital heart disease in neonates in China: a prospective study. Lancet. 2014 Aug 30;384(9945):747-54.
3. de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;Jan 8;338.
4. Ewer A et al. Pulse Oximetry Screening for Congenital Heart Defects in Newborn Infants (Pulseox): A Test Accuracy Study. Lancet. 2011 Aug 27;378(9793):785-94.
5. Taenzer AH et al. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology. 2010:112(2):282-287.
6. Taenzer A et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
7. McGrath SP et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
8. Estimate: Masimo data on file.
9. http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.