Globe Newswire11.15.18
CHF Solutions Inc. has entered into a collaboration agreement with NI Medical to explore potential clinical synergies between the company’s Aquadex FlexFlow System and NI Medical’s NICaS Hemodynamic Navigator to assess patient’s fluid volume status and assist by informing on the management of a fluid overloaded patient.
The collaboration will initiate with a small patient evaluation when combining the Aquadex FlexFlow system with NI Medical’s, already U.S. Food and Drug Administration 510k-cleared, NICaS Hemodynamic Navigator in clinical practice. The two companies intend to evaluate the potential benefits of utilizing the cardiovascular, respiratory and fluid diagnostic parameters from NI Medical’s NICaS Hemodynamic Navigator to better understand fluid volume and hemodynamic parameters in patients during an ultrafiltration procedure using the Aquadex FlexFlow System.
“We are dedicated to providing physicians with access to solutions that help them more effectively treat fluid overloaded patients”, said John Erb, chairman and CEO of CHF Solutions. “We believe that the collaboration with NI Medical allows us to assess the technology and provide a potential option to physicians to improve the treatment of patients with fluid overload when diuretics have failed.”
CHF Solutions Inc. is a medical device company focused on commercializing the Aquadex FlexFlow system for Aquapheresis therapy. The Aquadex FlexFlow system is indicated for temporary (up to eight hours) ultrafiltration treatment of patients with fluid overload who have failed diuretic therapy and extended (longer than eight hours) ultrafiltration treatment of patients with fluid overload who have failed diuretic therapy and require hospitalization. All treatments must be administered by a healthcare provider, under physician prescription, both of whom having received training in extracorporeal therapies. The company's mission is to predict, measure, and control patient fluid balance through science, collaboration, and innovative medical technology. CHF Solutions is a Delaware corporation headquartered in Minneapolis, Minn., with wholly owned subsidiaries in Australia and Ireland. The company has been listed on the NASDAQ Capital Market since February 2012.
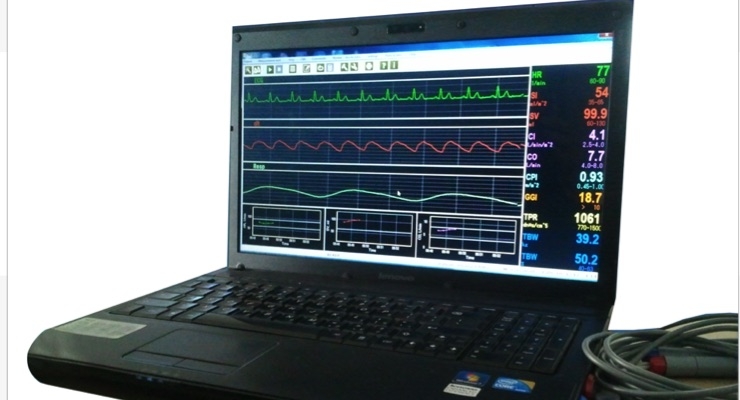
NI Medical is medical device company focused on commercializing the NICaS Hemodynamic Navigator. The NICaS Hemodynamic Navigator is a non-invasive hemodynamic monitor designed for continuous measuring of cardiac output, peripheral resistance, and body fluids. The system is intended to help improve treatment of congestive heart failure, hypertensive and dialysis patients. The system is designed to identify a patient’s hemodynamic state (vasoconstricted/ vasodilated) and volume status (hypovolemia/ hypervolemia). NI Medical is an Israeli corporation headquartered in Petach Tikva, Israel.
The collaboration will initiate with a small patient evaluation when combining the Aquadex FlexFlow system with NI Medical’s, already U.S. Food and Drug Administration 510k-cleared, NICaS Hemodynamic Navigator in clinical practice. The two companies intend to evaluate the potential benefits of utilizing the cardiovascular, respiratory and fluid diagnostic parameters from NI Medical’s NICaS Hemodynamic Navigator to better understand fluid volume and hemodynamic parameters in patients during an ultrafiltration procedure using the Aquadex FlexFlow System.
“We are dedicated to providing physicians with access to solutions that help them more effectively treat fluid overloaded patients”, said John Erb, chairman and CEO of CHF Solutions. “We believe that the collaboration with NI Medical allows us to assess the technology and provide a potential option to physicians to improve the treatment of patients with fluid overload when diuretics have failed.”
CHF Solutions Inc. is a medical device company focused on commercializing the Aquadex FlexFlow system for Aquapheresis therapy. The Aquadex FlexFlow system is indicated for temporary (up to eight hours) ultrafiltration treatment of patients with fluid overload who have failed diuretic therapy and extended (longer than eight hours) ultrafiltration treatment of patients with fluid overload who have failed diuretic therapy and require hospitalization. All treatments must be administered by a healthcare provider, under physician prescription, both of whom having received training in extracorporeal therapies. The company's mission is to predict, measure, and control patient fluid balance through science, collaboration, and innovative medical technology. CHF Solutions is a Delaware corporation headquartered in Minneapolis, Minn., with wholly owned subsidiaries in Australia and Ireland. The company has been listed on the NASDAQ Capital Market since February 2012.
NI Medical is medical device company focused on commercializing the NICaS Hemodynamic Navigator. The NICaS Hemodynamic Navigator is a non-invasive hemodynamic monitor designed for continuous measuring of cardiac output, peripheral resistance, and body fluids. The system is intended to help improve treatment of congestive heart failure, hypertensive and dialysis patients. The system is designed to identify a patient’s hemodynamic state (vasoconstricted/ vasodilated) and volume status (hypovolemia/ hypervolemia). NI Medical is an Israeli corporation headquartered in Petach Tikva, Israel.