Ethicon Inc. 11.05.18
Ethicon,* part of Johnson & Johnson Medical Devices Companies,** and a global leader in bariatric surgery, announced the launch of the Ethicon Bariatric Revision Surgical Solution, a new system for bariatric revision surgery that encompasses a portfolio of products and professional education and training for managing the challenges of reoperations.
Bariatric surgery has been shown to be the most effective long-term treatment for the disease of severe obesity,1 but in certain instances, patients may require revision surgery or a new treatment modality due to weight recidivism, changes in anatomy, inadequate weight loss or co-morbidity reduction or complications.2 Due to the nature and chronicity of the disease of obesity, lifelong management is required.
“When reoperations are required, they can be challenging due to the unpredictable nature and complexity of variables to which the surgeon may be confronted during surgery. These include such issues as adhesions, tissue type and thickness, unexpected or disruptive bleeding, and difficult access to the patient’s anatomy. This new portfolio of products and education from Ethicon helps the surgeon manage this unpredictability to the benefit of the patient,” said Alan Wittgrove, M.D., FACS, FASMBS, an Ethicon consultant and medical director at the Wittgrove Bariatric Center in Del Mar, Calif.
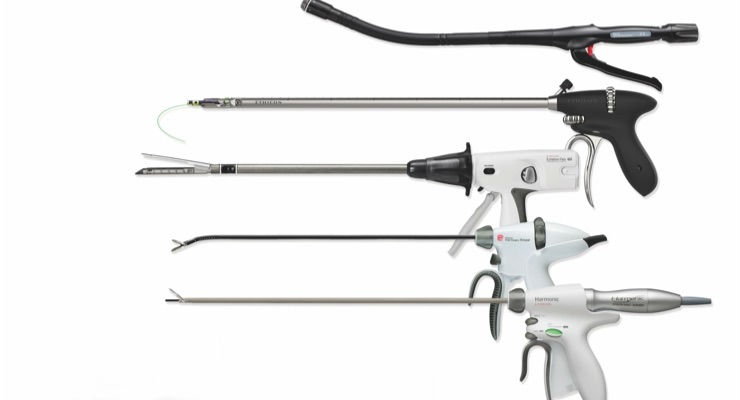
Ethicon’s Bariatric Revision Surgical Solution includes a product portfolio designed to help surgeons perform bariatric revision surgery with precision, efficiency and consistency. It includes the HARMONIC HD 1000i Shears, an advanced energy device for precise dissection and secure sealing of tissue, the ECHELON FLEX GST System for exceptional staple line integrity across a wide range of tissue thickness,3 STRATAFIX Knotless Tissue Control Devices for suturing with consistent tension control, and SURGICEL SNoW Absorbable Hemostat for enhanced hemostatic performance, conformability and tissue adherence.4
Education and training also play a critical role in preparing bariatric surgeons for revision surgery and Ethicon is filling this important need with bariatric revision courses, symposia and interactive learning experiences offered through the Johnson & Johnson Institute, which brings together 26 professional education facilities and a network of online education and collaborative partnerships across multiple specialties.
“Over the last 20 years, Ethicon has invested significantly in the treatment of obesity and related diseases,” said Michael del Prado, Ethicon Company group chairman. “Based on this legacy of leadership and collaboration, bariatric surgeons, healthcare professionals and patients can rely on Ethicon to provide comprehensive solutions that help them effectively manage the challenges of primary bariatric surgery and revision surgery, and the disease of obesity, both inside and outside the operating room.”
The Johnson & Johnson Medical Devices Companies’ purpose is to reach more patients and restore more lives. Having advanced patient care for more than a century, these companies represent a breadth of products, services, programs, and research and development capabilities in surgical technology, orthopedics, interventional, and specialty solutions with an offering directed at delivering clinical and economic value to health care systems worldwide.
From creating the first sutures, to revolutionizing surgery with minimally invasive procedures, Ethicon, part of the Johnson & Johnson Medical Devices Companies, has made significant contributions to surgery for more than 60 years. Through Ethicon’s surgical technologies and solutions include sutures, staplers, energy devices, trocars and hemostats.
* Ethicon represents the products and services of Ethicon Inc., Ethicon Endo-Surgery LLC, and certain of their affiliates.
** The Johnson & Johnson Medical Devices Companies comprise the surgery, orthopedics, and interventional solutions businesses within Johnson & Johnson’s Medical Devices segment.
References
1. Weiner R. (2010). Indications and principles of metabolic surgery. Der Chirug. 81(4):379-394. https://www.ncbi.nlm.nih.gov/pubmed/20361370
2. Brethauer S, Kothari S, Sudan R, et al. (2014). Systematic review on reoperative bariatric surgery, American Society for Metabolic and Bariatric Surgery Revision Task Force. Surg Obes Relat Dis. 10(5):952-972. https://asmbs.org/resources/systematic-review-on-reoperative
3. The ECHELON FLEX GST System was designed and tested to meet rigorous design requirements for staple line integrity. The GST System accommodates a tissue thickness range of 1.0mm to 4.0mm (measured at 8g/mm2, tissue comfortably compressed to closed staple height during firing per IFU) while the Medtronic Tri-StapleTM portfolio is intended for a tissue thickness range of 0.75mm to 3.0mm (per IFU & materials downloaded from Medtronic website on Nov 16, 2016).
4. https://www.ethicon.com/na/products/adjunctive-hemostasis/absorbable-hemostats/surgicel-snow-absorbable-hemostat
Bariatric surgery has been shown to be the most effective long-term treatment for the disease of severe obesity,1 but in certain instances, patients may require revision surgery or a new treatment modality due to weight recidivism, changes in anatomy, inadequate weight loss or co-morbidity reduction or complications.2 Due to the nature and chronicity of the disease of obesity, lifelong management is required.
“When reoperations are required, they can be challenging due to the unpredictable nature and complexity of variables to which the surgeon may be confronted during surgery. These include such issues as adhesions, tissue type and thickness, unexpected or disruptive bleeding, and difficult access to the patient’s anatomy. This new portfolio of products and education from Ethicon helps the surgeon manage this unpredictability to the benefit of the patient,” said Alan Wittgrove, M.D., FACS, FASMBS, an Ethicon consultant and medical director at the Wittgrove Bariatric Center in Del Mar, Calif.
Ethicon’s Bariatric Revision Surgical Solution includes a product portfolio designed to help surgeons perform bariatric revision surgery with precision, efficiency and consistency. It includes the HARMONIC HD 1000i Shears, an advanced energy device for precise dissection and secure sealing of tissue, the ECHELON FLEX GST System for exceptional staple line integrity across a wide range of tissue thickness,3 STRATAFIX Knotless Tissue Control Devices for suturing with consistent tension control, and SURGICEL SNoW Absorbable Hemostat for enhanced hemostatic performance, conformability and tissue adherence.4
Education and training also play a critical role in preparing bariatric surgeons for revision surgery and Ethicon is filling this important need with bariatric revision courses, symposia and interactive learning experiences offered through the Johnson & Johnson Institute, which brings together 26 professional education facilities and a network of online education and collaborative partnerships across multiple specialties.
“Over the last 20 years, Ethicon has invested significantly in the treatment of obesity and related diseases,” said Michael del Prado, Ethicon Company group chairman. “Based on this legacy of leadership and collaboration, bariatric surgeons, healthcare professionals and patients can rely on Ethicon to provide comprehensive solutions that help them effectively manage the challenges of primary bariatric surgery and revision surgery, and the disease of obesity, both inside and outside the operating room.”
The Johnson & Johnson Medical Devices Companies’ purpose is to reach more patients and restore more lives. Having advanced patient care for more than a century, these companies represent a breadth of products, services, programs, and research and development capabilities in surgical technology, orthopedics, interventional, and specialty solutions with an offering directed at delivering clinical and economic value to health care systems worldwide.
From creating the first sutures, to revolutionizing surgery with minimally invasive procedures, Ethicon, part of the Johnson & Johnson Medical Devices Companies, has made significant contributions to surgery for more than 60 years. Through Ethicon’s surgical technologies and solutions include sutures, staplers, energy devices, trocars and hemostats.
* Ethicon represents the products and services of Ethicon Inc., Ethicon Endo-Surgery LLC, and certain of their affiliates.
** The Johnson & Johnson Medical Devices Companies comprise the surgery, orthopedics, and interventional solutions businesses within Johnson & Johnson’s Medical Devices segment.
References
1. Weiner R. (2010). Indications and principles of metabolic surgery. Der Chirug. 81(4):379-394. https://www.ncbi.nlm.nih.gov/pubmed/20361370
2. Brethauer S, Kothari S, Sudan R, et al. (2014). Systematic review on reoperative bariatric surgery, American Society for Metabolic and Bariatric Surgery Revision Task Force. Surg Obes Relat Dis. 10(5):952-972. https://asmbs.org/resources/systematic-review-on-reoperative
3. The ECHELON FLEX GST System was designed and tested to meet rigorous design requirements for staple line integrity. The GST System accommodates a tissue thickness range of 1.0mm to 4.0mm (measured at 8g/mm2, tissue comfortably compressed to closed staple height during firing per IFU) while the Medtronic Tri-StapleTM portfolio is intended for a tissue thickness range of 0.75mm to 3.0mm (per IFU & materials downloaded from Medtronic website on Nov 16, 2016).
4. https://www.ethicon.com/na/products/adjunctive-hemostasis/absorbable-hemostats/surgicel-snow-absorbable-hemostat