Business Wire10.29.18
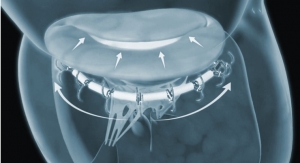
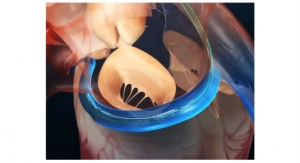
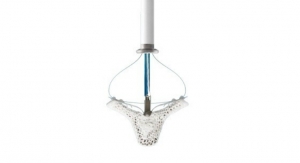
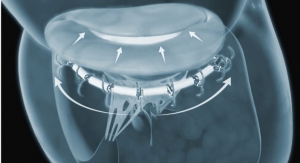
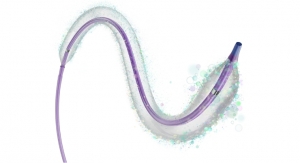
Advanced Bifurcation Systems (ABS), a clinical-stage medical device company with a patented bifurcation stenting platform to standardize the treatment of all bifurcation lesions in coronary angioplasties, has unveiled a platform featuring the only modular self-aligning stent technology to evenly scaffold bifurcations.
“Current statistics for bifurcation stenting show the highest stenting failure rates resulting in repeat revascularization procedures,” said Mehran Khorsandi, M.D., interventional cardiologist and member of the HARTS stent team, Cedars-Sinai Medical Center, co-founder and CMO of ABS. “In the absence of an effective reproducible technology for addressing left main bifurcation disease, most patients are referred for open-heart surgery. The ABS technology overcomes the limitations of current approaches while simplifying the procedure.”
The ABS platform is a unique single system addressing main and side branches offering a disruptive bifurcation stenting technique that:
“Our stenting platform reduces the need for repeat procedures and enables effective bifurcation stenting treatment,” said ABS CEO Charles Laverty. “This procedure addresses a significant portion of patients who otherwise would be referred to open-heart surgery.”
ABS has filed a pre-submission with the U.S. Food and Drug Administration to define its clinical pathway leading to pre-market approval. The company also is defining its regulatory and clinical strategy for Europe to obtain a CE Mark.
Advanced Bifurcation Systems is a clinical-stage medical device company developing a stenting platform for simple treatment of bifurcation lesions in coronary angioplasties. The company is based in Newport Beach, Calif.
“Current statistics for bifurcation stenting show the highest stenting failure rates resulting in repeat revascularization procedures,” said Mehran Khorsandi, M.D., interventional cardiologist and member of the HARTS stent team, Cedars-Sinai Medical Center, co-founder and CMO of ABS. “In the absence of an effective reproducible technology for addressing left main bifurcation disease, most patients are referred for open-heart surgery. The ABS technology overcomes the limitations of current approaches while simplifying the procedure.”
The ABS platform is a unique single system addressing main and side branches offering a disruptive bifurcation stenting technique that:
- Uses bifurcating stents deployed on a Mother/Daughter (MD) monorail to branch and self-align covering bifurcations with no gaps;
- Uses a dual-catheter system to move stents into position in one continuous process; and
- Eliminates gaps or overlaps and tangling of wires associated with current stent installations.
“Our stenting platform reduces the need for repeat procedures and enables effective bifurcation stenting treatment,” said ABS CEO Charles Laverty. “This procedure addresses a significant portion of patients who otherwise would be referred to open-heart surgery.”
ABS has filed a pre-submission with the U.S. Food and Drug Administration to define its clinical pathway leading to pre-market approval. The company also is defining its regulatory and clinical strategy for Europe to obtain a CE Mark.
Advanced Bifurcation Systems is a clinical-stage medical device company developing a stenting platform for simple treatment of bifurcation lesions in coronary angioplasties. The company is based in Newport Beach, Calif.