U.S. Food and Drug Administration10.16.18
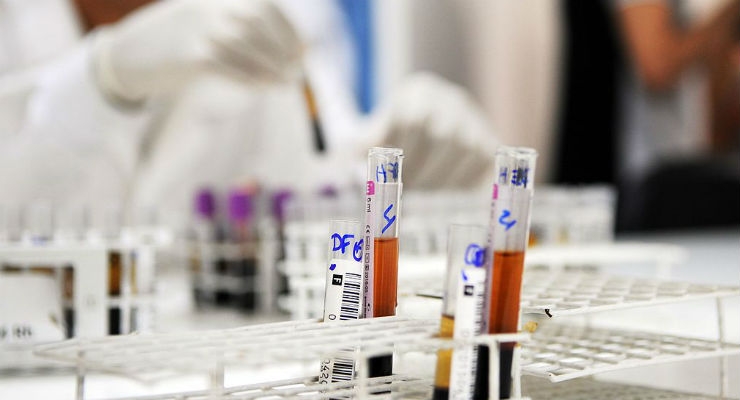
The U.S. Food and Drug Administration approved ID CORE XT, a molecular-based assay used in blood transfusion medicine to help determine blood compatibility. The assay can be used to determine blood donor and patient non-ABO red blood cell (RBC) types. ID CORE XT is the second molecular assay approved for use in transfusion medicine, and the first to report genotypes as final results.
“The approval of the ID CORE XT Test can streamline blood compatibility testing and provides an additional alternative to testing blood with antisera,” said Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research. “We know that DNA testing holds great promise—to provide more informative, accurate and cost-effective methods that can enhance patient care.”
Human blood can be classified into different groups based on the antigens on the surfaces of red blood cells. In addition to the ABO blood group antigens, the presence or absence of other specific blood group antigens can be important when matching blood for transfusions since some people develop antibodies to non-ABO antigens. People who receive repeated blood transfusions, such as individuals with sickle cell disease, are more likely to develop these antibodies. If red blood cells with poorly matched non-ABO antigens are transfused, red blood cell destruction and a transfusion reaction can occur in a transfusion recipient.
Traditionally, red blood cell antigens have been identified using serological methods that involve the use of antisera, a blood serum that contains antibodies for testing. Serologic testing presents limitations and certain antisera may be scarce or unavailable.
A study was conducted to compare the typing results of the ID CORE XT Test with licensed serological reagents, the first FDA-approved molecular assay, and DNA sequencing tests. The results demonstrated comparable performance between the methods.
The approval of the ID CORE XT Test was granted to Progenika Biopharma S.A., a Grifols company.
“The approval of the ID CORE XT Test can streamline blood compatibility testing and provides an additional alternative to testing blood with antisera,” said Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research. “We know that DNA testing holds great promise—to provide more informative, accurate and cost-effective methods that can enhance patient care.”
Human blood can be classified into different groups based on the antigens on the surfaces of red blood cells. In addition to the ABO blood group antigens, the presence or absence of other specific blood group antigens can be important when matching blood for transfusions since some people develop antibodies to non-ABO antigens. People who receive repeated blood transfusions, such as individuals with sickle cell disease, are more likely to develop these antibodies. If red blood cells with poorly matched non-ABO antigens are transfused, red blood cell destruction and a transfusion reaction can occur in a transfusion recipient.
Traditionally, red blood cell antigens have been identified using serological methods that involve the use of antisera, a blood serum that contains antibodies for testing. Serologic testing presents limitations and certain antisera may be scarce or unavailable.
A study was conducted to compare the typing results of the ID CORE XT Test with licensed serological reagents, the first FDA-approved molecular assay, and DNA sequencing tests. The results demonstrated comparable performance between the methods.
The approval of the ID CORE XT Test was granted to Progenika Biopharma S.A., a Grifols company.