Business Wire10.03.18
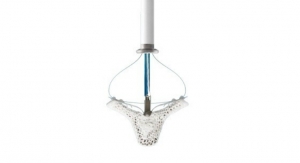
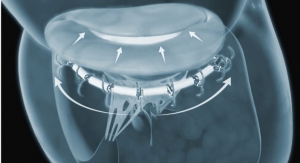
Acessa Health Inc., developer of the Acessa System, announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) and U.S. commercial availability of the Acessa System’s most advanced and proprietary technology, the Acessa ProVu System. A fully-integrated radiofrequency ablation, ultrasound visualization, and guidance mapping system, Acessa ProVu enables physicians to effectively and safely treat women with symptomatic uterine fibroids.
The Acessa Procedure is specifically designed to treat women suffering from uterine fibroids and is clinically proven with long-term data as a safe alternative to hysterectomy. Nearly 3,000 procedures have been performed to-date. Acessa ProVu is the third generation of Acessa’s minimally invasive, laparoscopic radiofrequency ablation solution—and is the first and only solution with a category 1 CPT reimbursement code and widely established coverage.
“The FDA clearance of Acessa ProVu is a significant step forward in terms of offering patients more minimally invasive options to address their fibroids,” said Dr. Regina Benjamin, M.D., 18th Surgeon General of the United States.
Uterine fibroids affect 70–80 percent of women under the age of 55. In the U.S., hysterectomy has historically been the primary treatment for fibroids. As a result, hysterectomy is the second most common surgery performed on reproductive-aged women. The Acessa Procedure is a safe, effective, and minimally invasive alternative. The procedure requires no cutting or suturing of uterine tissue and patients typically go home the same day, experiencing minimal discomfort and a rapid return to normal activities.
“The Acessa System is the most utilized and advanced procedure for laparoscopic radiofrequency ablation of symptomatic uterine fibroids. After personally treating over 300 patients to-date, I am thrilled to incorporate the Acessa ProVu System in my practice,” said Dr. Abraham Shashoua, Director of Minimally Invasive and Robotic Gynecologic Surgery at Advocate Illinois Masonic Medical Center.
“We have seen substantial benefits of laparoscopic radiofrequency ablation for the treatment of symptomatic fibroids,” said David Eisenstein, M.D. the Director of Henry Ford Health System’s Center of Excellence in Minimally Invasive Gynecology. “As the Principal Investigator of TRUST and Co-Investigator of COMPARE-UF, two large fibroid treatment studies, we are excited for Acessa ProVu, which integrates imaging and energy into one system which will improve surgeon efficiency and accuracy in providing this minimally invasive option to patients.”
“Acessa Health is delighted to usher in our next phase of innovation with the introduction of Acessa ProVu,” said Kim Rodriguez, president and CEO of Acessa Health Inc. “Each phase of innovation—the original Acessa System, Acessa Guidance System and now the Acessa ProVu System—represent a significant improvement in performance and the opportunity to further support our physician partners who help women suffering from uterine fibroid symptoms.”
The Acessa Procedure is specifically designed to treat women suffering from uterine fibroids and is clinically proven with long-term data as a safe alternative to hysterectomy. Nearly 3,000 procedures have been performed to-date. Acessa ProVu is the third generation of Acessa’s minimally invasive, laparoscopic radiofrequency ablation solution—and is the first and only solution with a category 1 CPT reimbursement code and widely established coverage.
“The FDA clearance of Acessa ProVu is a significant step forward in terms of offering patients more minimally invasive options to address their fibroids,” said Dr. Regina Benjamin, M.D., 18th Surgeon General of the United States.
Uterine fibroids affect 70–80 percent of women under the age of 55. In the U.S., hysterectomy has historically been the primary treatment for fibroids. As a result, hysterectomy is the second most common surgery performed on reproductive-aged women. The Acessa Procedure is a safe, effective, and minimally invasive alternative. The procedure requires no cutting or suturing of uterine tissue and patients typically go home the same day, experiencing minimal discomfort and a rapid return to normal activities.
“The Acessa System is the most utilized and advanced procedure for laparoscopic radiofrequency ablation of symptomatic uterine fibroids. After personally treating over 300 patients to-date, I am thrilled to incorporate the Acessa ProVu System in my practice,” said Dr. Abraham Shashoua, Director of Minimally Invasive and Robotic Gynecologic Surgery at Advocate Illinois Masonic Medical Center.
“We have seen substantial benefits of laparoscopic radiofrequency ablation for the treatment of symptomatic fibroids,” said David Eisenstein, M.D. the Director of Henry Ford Health System’s Center of Excellence in Minimally Invasive Gynecology. “As the Principal Investigator of TRUST and Co-Investigator of COMPARE-UF, two large fibroid treatment studies, we are excited for Acessa ProVu, which integrates imaging and energy into one system which will improve surgeon efficiency and accuracy in providing this minimally invasive option to patients.”
“Acessa Health is delighted to usher in our next phase of innovation with the introduction of Acessa ProVu,” said Kim Rodriguez, president and CEO of Acessa Health Inc. “Each phase of innovation—the original Acessa System, Acessa Guidance System and now the Acessa ProVu System—represent a significant improvement in performance and the opportunity to further support our physician partners who help women suffering from uterine fibroid symptoms.”