NuCryo Vascular LLC09.14.18
NuCryo Vascular has signed a commercialization agreement with Lokai Medical, a specialty distributor of coronary/peripheral and interventional devices, to distribute the PolarCath Balloon Dilatation System in the United States.
“We are very excited to partner with Lokai Medical and the company’s experienced sales team to help commercialize the PolarCath Balloon Dilatation System,” said Kevin Beedon, general manager and executive vice president of NuCryo Vascular. "NuCryo grew sales by 36 percent in 2017 vs. 2016 with an extremely small but direct sales team. The commercialization support and clinical expertise of Lokai Medical will help NuCryo continue its double-digit growth in the expanded markets we can now serve.”
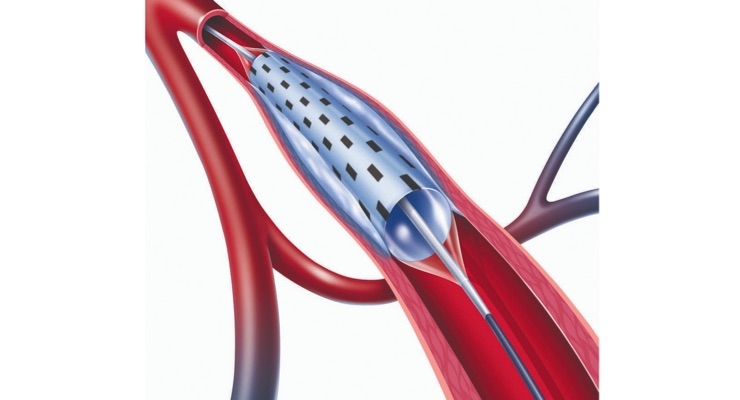
Balloon Cryoplasty has been shown in clinical studies and in daily procedures to be an important option for treating peripheral arterial disease. The controlled cooling of the plaque and artery wall provides three potential benefits: uniform vessel dilation with less vessel trauma, reduced vessel wall recoil and induction of apoptosis, which promotes the natural cell death of the smooth muscle cells that otherwise proliferate to cause restenosis. The PolarCath Cryoplasty system was invented by James Joye, M.D., an interventional cardiologist in Mountain View, Calif., who is a pioneer in the development of medical devices to treat peripheral arterial disease.
“I am very excited for the next-generation reusable Cryoplasty inflation device that NuCryo has re-engineered and brought back to market,” said Jonathan Aliota, M.D., interventional cardiologist from Houston, Texas. “As a previous Cryoplasty user with Boston Scientific, I recently reincorporated it back into my peripheral treatment algorithm and have been extremely happy with the results. In addition, the cost, ease of use, and time savings as compared to other balloons on the market have been well received at my facility.”
The PolarCath Peripheral Dilatation System received U.S. Food and Drug Administration (FDA) clearance to dilate stenosis in the peripheral vasculature (iliac, femoral, popliteal, infrapopliteal, renal, and subclavian arteries) and for the treatment of obstructive lesions of polytetrafluoroethylene (PTFE) access grafts or arteriovenous dialysis fistulae. The PolarCath System is also indicated for post-deployed stent expansion of self-expanding peripheral vascular stents.
The PolarCath system, which received its first FDA clearance in 2002, was developed by CryoVascular Inc., a company formed by Joye and collaborators. In 2005, CryoVascular was acquired by Boston Scientific, which discontinued manufacturing PolarCath at the end of 2012. NuCryo Vascular created in 2014 for the purpose of acquiring PolarCath and resuming manufacturing of the device. NuCryo Vascular has since re-engineered the device and received FDA approval of the non-sterile inflation device in 2015.
NuCryo Vascular LLC is a privately held company that manufactures, markets, and sells the PolarCath (Cryoplasty) Balloon Dilatation System.
Lokai Medical is a specialty distributor of coronary/peripheral and interventional devices throughout the United States. Lokai Medical is based in Phoenix, Ariz.
“We are very excited to partner with Lokai Medical and the company’s experienced sales team to help commercialize the PolarCath Balloon Dilatation System,” said Kevin Beedon, general manager and executive vice president of NuCryo Vascular. "NuCryo grew sales by 36 percent in 2017 vs. 2016 with an extremely small but direct sales team. The commercialization support and clinical expertise of Lokai Medical will help NuCryo continue its double-digit growth in the expanded markets we can now serve.”
Balloon Cryoplasty has been shown in clinical studies and in daily procedures to be an important option for treating peripheral arterial disease. The controlled cooling of the plaque and artery wall provides three potential benefits: uniform vessel dilation with less vessel trauma, reduced vessel wall recoil and induction of apoptosis, which promotes the natural cell death of the smooth muscle cells that otherwise proliferate to cause restenosis. The PolarCath Cryoplasty system was invented by James Joye, M.D., an interventional cardiologist in Mountain View, Calif., who is a pioneer in the development of medical devices to treat peripheral arterial disease.
“I am very excited for the next-generation reusable Cryoplasty inflation device that NuCryo has re-engineered and brought back to market,” said Jonathan Aliota, M.D., interventional cardiologist from Houston, Texas. “As a previous Cryoplasty user with Boston Scientific, I recently reincorporated it back into my peripheral treatment algorithm and have been extremely happy with the results. In addition, the cost, ease of use, and time savings as compared to other balloons on the market have been well received at my facility.”
The PolarCath Peripheral Dilatation System received U.S. Food and Drug Administration (FDA) clearance to dilate stenosis in the peripheral vasculature (iliac, femoral, popliteal, infrapopliteal, renal, and subclavian arteries) and for the treatment of obstructive lesions of polytetrafluoroethylene (PTFE) access grafts or arteriovenous dialysis fistulae. The PolarCath System is also indicated for post-deployed stent expansion of self-expanding peripheral vascular stents.
The PolarCath system, which received its first FDA clearance in 2002, was developed by CryoVascular Inc., a company formed by Joye and collaborators. In 2005, CryoVascular was acquired by Boston Scientific, which discontinued manufacturing PolarCath at the end of 2012. NuCryo Vascular created in 2014 for the purpose of acquiring PolarCath and resuming manufacturing of the device. NuCryo Vascular has since re-engineered the device and received FDA approval of the non-sterile inflation device in 2015.
NuCryo Vascular LLC is a privately held company that manufactures, markets, and sells the PolarCath (Cryoplasty) Balloon Dilatation System.
Lokai Medical is a specialty distributor of coronary/peripheral and interventional devices throughout the United States. Lokai Medical is based in Phoenix, Ariz.