Business Wire08.09.18
Endologix Inc., a developer and marketer of treatments for aortic disorders, announced one-year results from the LUCY (Evaluation of FemaLes who are Underrepresented Candidates for Abdominal Aortic AneurYsm Repair) registry. The LUCY study is the first to prospectively evaluate endovascular aneurysm repair (EVAR) outcomes in women who have more complex aortic anatomy and, subsequently, have worse reported outcomes than men undergoing EVAR.1 The results of the LUCY one-year data expand on the 30-day results presented last year, showing that at least 28 percent more women are eligible for minimally-invasive EVAR when using the Ovation Abdominal Stent Graft System than when using other EVAR systems.2
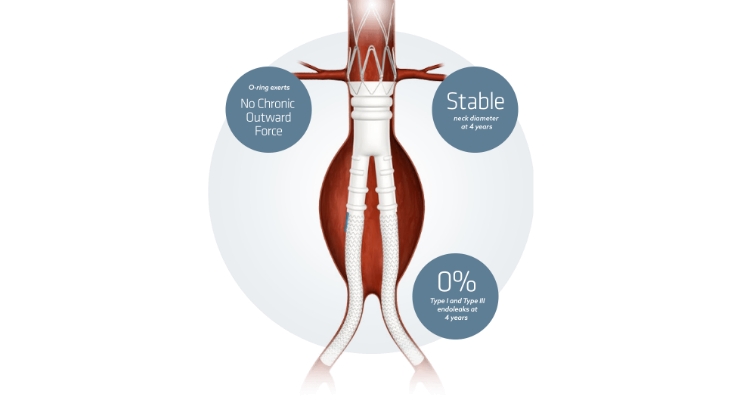
The LUCY study is a prospective, consecutively enrolling, non-randomized, multi-center, post-market registry evaluating the Ovation System for the endovascular treatment of abdominal aortic aneurysms (AAA) in women.3 The study enrolled a total of 225 patients, including 76 females in the treatment group and 149 males in the control group, at 39 sites in the United States. The primary endpoint of the study was the 30-day Major Adverse Event (MAE) rate and secondary endpoints including serious and non-serious adverse events through one year.
The one-year LUCY data showed that, despite having more complex anatomy at the time of the index procedure than men, women had similar outcomes to men following treatment with the ultra-low profile (14F) Ovation device:
Jennifer Ash, M.D., Christie Clinic Vein and Vascular Center and assistant clinical professor of Surgery, University of Illinois College of Medicine in Champaign, said, “Women have specific anatomical challenges in iliac access and proximal aortic neck morphology and have historically had worse outcomes from EVAR than men. The results of the LUCY study suggest that the unique features of the low-profile Ovation system may overcome these challenges and achieve similar outcomes in men and women.”4,5
Endologix CEO John Onopchenko commented, “We are excited to have completed follow-up on the LUCY study and are pleased that the one-year results suggest, for the first time in a prospective study, that female AAA patients can be treated with EVAR as effectively as men, when using the Ovation System. LUCY’s evidence provides physicians with new information to confidently engage female patients with prospectively derived outcomes previously realized only by male patients.”
The LUCY study was led by an advisory board whose members include: Chairperson Jennifer Ash, M.D., Christie Clinic Vein and Vascular Center and assistant clinical professor of Surgery, University of Illinois College of Medicine in Champaign; Venita Chandra, M.D., clinical assistant professor of Surgery-Vascular Surgery, Stanford School of Medicine; Monica Hunter, M.D., Birmingham Heart Clinic at St. Vincent’s Birmingham; Eva Rzucidlo, M.D., McLeod Vascular and associate professor of Surgery, Geisel School of Medicine, Dartmouth; and Ageliki Vouyouka, M.D., associate professor of Surgery and Radiology, Mount Sinai Hospital.
Endologix Inc. develops and manufactures minimally invasive treatments for aortic disorders. The company's focus is endovascular stent grafts for the treatment of abdominal aortic aneurysms (AAA). AAA is a weakening of the wall of the aorta, the largest artery in the body, resulting in a balloon-like enlargement. Once AAA develops, it continues to enlarge and, if left untreated, becomes increasingly susceptible to rupture. The overall patient mortality rate for ruptured AAA is approximately 80 percent, making it a leading cause of death in the United States. www.endologix.com.
References
1. Pinar Ulug, PhD, et al. www.thelancet.com Published online April 25, 2017 Sweet, M, M.D., et al. J Vasc Surg 2011; 54:931-7.
2. Analysis based on available data from the LUCY Study female cohort (72 out of 76) and on comparisons with grafts ranging from 18F – 21F OD manufactured by global market leaders. Data extracted on May 1, 2017. The Ovation Abdominal Stent Graft System has not been studied in a head-to-head clinical study against other EVAR devices for outcomes in women.
3. Two percent of patients had vascular characteristics beyond the FDA-approved anatomic IFU. Safety and effectiveness, of Ovation when used off-IFU, have not been established.
4. Mehta M, Byrne WJ, Robinson H, et al. “Women derive less benefit from elective endovascular aneurysm repair than men.” J Vasc Surg. Apr 2012;55(4):906-913.
5. Lowry, D, et al. “Sex-related Outcome Inequalities in Endovascular Aneurysm Repair.” Eur J Vasc Endovasc Surg (2016) 1-8.
The LUCY study is a prospective, consecutively enrolling, non-randomized, multi-center, post-market registry evaluating the Ovation System for the endovascular treatment of abdominal aortic aneurysms (AAA) in women.3 The study enrolled a total of 225 patients, including 76 females in the treatment group and 149 males in the control group, at 39 sites in the United States. The primary endpoint of the study was the 30-day Major Adverse Event (MAE) rate and secondary endpoints including serious and non-serious adverse events through one year.
The one-year LUCY data showed that, despite having more complex anatomy at the time of the index procedure than men, women had similar outcomes to men following treatment with the ultra-low profile (14F) Ovation device:
- Freedom from AAA-related morality: 100 percent in women vs. 98.6 percent in men
- Freedom from reintervention for Type 1a endoleak: 98.6 percent in women vs. 97.9 percent in men
- Freedom from rupture: 100 percent in both women and men
- Freedom from conversion: 100 percent in both women and men
- Freedom from all device-related reintervention: 97.2 percent in both women and men
Jennifer Ash, M.D., Christie Clinic Vein and Vascular Center and assistant clinical professor of Surgery, University of Illinois College of Medicine in Champaign, said, “Women have specific anatomical challenges in iliac access and proximal aortic neck morphology and have historically had worse outcomes from EVAR than men. The results of the LUCY study suggest that the unique features of the low-profile Ovation system may overcome these challenges and achieve similar outcomes in men and women.”4,5
Endologix CEO John Onopchenko commented, “We are excited to have completed follow-up on the LUCY study and are pleased that the one-year results suggest, for the first time in a prospective study, that female AAA patients can be treated with EVAR as effectively as men, when using the Ovation System. LUCY’s evidence provides physicians with new information to confidently engage female patients with prospectively derived outcomes previously realized only by male patients.”
The LUCY study was led by an advisory board whose members include: Chairperson Jennifer Ash, M.D., Christie Clinic Vein and Vascular Center and assistant clinical professor of Surgery, University of Illinois College of Medicine in Champaign; Venita Chandra, M.D., clinical assistant professor of Surgery-Vascular Surgery, Stanford School of Medicine; Monica Hunter, M.D., Birmingham Heart Clinic at St. Vincent’s Birmingham; Eva Rzucidlo, M.D., McLeod Vascular and associate professor of Surgery, Geisel School of Medicine, Dartmouth; and Ageliki Vouyouka, M.D., associate professor of Surgery and Radiology, Mount Sinai Hospital.
Endologix Inc. develops and manufactures minimally invasive treatments for aortic disorders. The company's focus is endovascular stent grafts for the treatment of abdominal aortic aneurysms (AAA). AAA is a weakening of the wall of the aorta, the largest artery in the body, resulting in a balloon-like enlargement. Once AAA develops, it continues to enlarge and, if left untreated, becomes increasingly susceptible to rupture. The overall patient mortality rate for ruptured AAA is approximately 80 percent, making it a leading cause of death in the United States. www.endologix.com.
References
1. Pinar Ulug, PhD, et al. www.thelancet.com Published online April 25, 2017 Sweet, M, M.D., et al. J Vasc Surg 2011; 54:931-7.
2. Analysis based on available data from the LUCY Study female cohort (72 out of 76) and on comparisons with grafts ranging from 18F – 21F OD manufactured by global market leaders. Data extracted on May 1, 2017. The Ovation Abdominal Stent Graft System has not been studied in a head-to-head clinical study against other EVAR devices for outcomes in women.
3. Two percent of patients had vascular characteristics beyond the FDA-approved anatomic IFU. Safety and effectiveness, of Ovation when used off-IFU, have not been established.
4. Mehta M, Byrne WJ, Robinson H, et al. “Women derive less benefit from elective endovascular aneurysm repair than men.” J Vasc Surg. Apr 2012;55(4):906-913.
5. Lowry, D, et al. “Sex-related Outcome Inequalities in Endovascular Aneurysm Repair.” Eur J Vasc Endovasc Surg (2016) 1-8.