Globe Newswire08.08.18
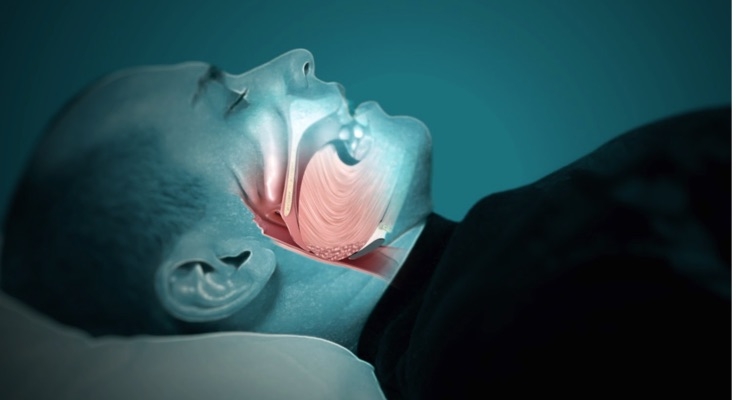
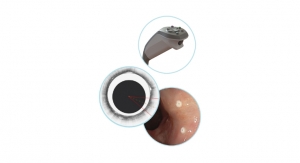
Inspire Medical Systems Inc., a medical technology company focused on the development and commercialization of minimally invasive solutions for patients with obstructive sleep apnea (OSA), announced that Japan’s Ministry of Health, Labour and Welfare has approved its Inspire therapy to treat moderate to severe OSA.
Inspire therapy is a closed-loop, minimally invasive solution that provides comfort and convenience, resulting in high compliance for patients with moderate to severe OSA. The safety and efficacy of Inspire therapy is supported by a significant body of clinical data, which includes a publication in the New England Journal of Medicine and more than 50 peer-reviewed publications. Inspire obtained CE Mark for its Inspire therapy in 2010 and U.S. Food and Drug Administration approval in 2014. To date, physicians have treated more than 3,000 patients worldwide with Inspire therapy.
“With this approval for our Inspire therapy, we are able to provide patients and physicians in Japan with a new alternative for the treatment of moderate to severe OSA,” said Tim Herbert, president and CEO of Inspire Medical Systems. “Our Inspire therapy is supported by a strong body of evidence with more than five years of clinical experience demonstrating the efficacy and safety of our therapy for patients and physicians. We look forward to commercializing our Inspire therapy in Japan.”
Recent published data estimates that 9 percent of Japanese men and nearly 3 percent of Japanese women are diagnosed with OSA. Further reports indicate that approximately 9 million Japanese have an Apnea Hypopnea Index (AHI) greater than 15, which qualifies them at least as moderate OSA patients. Inspire will now seek reimbursement coverage for its therapy in Japan. The reimbursement process will require significant time and interactions with the authorities in Japan. As a result, along with the planned organizational development and physician training, Inspire intends to launch its therapy in Japan in the second half of 2019.
“We would like to thank Cobridge Co. Ltd., a regulatory affairs consulting firm that provides comprehensive product registration services for medical products in Japan, for their efforts and expertise in assisting us to gain approval in Japan,” said Herbert.
Inspire Medical Systems is focused on the development and commercialization of minimally invasive solutions for patients with obstructive sleep apnea. Inspire Medical Systems’ proprietary Inspire therapy is the first and only U.S. Food and Drug Administration-approved neurostimulation technology that provides a safe and effective treatment for moderate to severe obstructive sleep apnea.
Inspire therapy is a closed-loop, minimally invasive solution that provides comfort and convenience, resulting in high compliance for patients with moderate to severe OSA. The safety and efficacy of Inspire therapy is supported by a significant body of clinical data, which includes a publication in the New England Journal of Medicine and more than 50 peer-reviewed publications. Inspire obtained CE Mark for its Inspire therapy in 2010 and U.S. Food and Drug Administration approval in 2014. To date, physicians have treated more than 3,000 patients worldwide with Inspire therapy.
“With this approval for our Inspire therapy, we are able to provide patients and physicians in Japan with a new alternative for the treatment of moderate to severe OSA,” said Tim Herbert, president and CEO of Inspire Medical Systems. “Our Inspire therapy is supported by a strong body of evidence with more than five years of clinical experience demonstrating the efficacy and safety of our therapy for patients and physicians. We look forward to commercializing our Inspire therapy in Japan.”
Recent published data estimates that 9 percent of Japanese men and nearly 3 percent of Japanese women are diagnosed with OSA. Further reports indicate that approximately 9 million Japanese have an Apnea Hypopnea Index (AHI) greater than 15, which qualifies them at least as moderate OSA patients. Inspire will now seek reimbursement coverage for its therapy in Japan. The reimbursement process will require significant time and interactions with the authorities in Japan. As a result, along with the planned organizational development and physician training, Inspire intends to launch its therapy in Japan in the second half of 2019.
“We would like to thank Cobridge Co. Ltd., a regulatory affairs consulting firm that provides comprehensive product registration services for medical products in Japan, for their efforts and expertise in assisting us to gain approval in Japan,” said Herbert.
Inspire Medical Systems is focused on the development and commercialization of minimally invasive solutions for patients with obstructive sleep apnea. Inspire Medical Systems’ proprietary Inspire therapy is the first and only U.S. Food and Drug Administration-approved neurostimulation technology that provides a safe and effective treatment for moderate to severe obstructive sleep apnea.