Business Wire07.25.18
Kurin Inc., inventor and manufacturer of the U.S. Food and Drug Administration (FDA) 510(k)-cleared Kurin blood culture collection sets, announced new clinical study data supporting the efficacy of their specimen diversion device for blood culture collection. Kurin offers the only FDA 510(k)-cleared blood culture diversion product on the market.
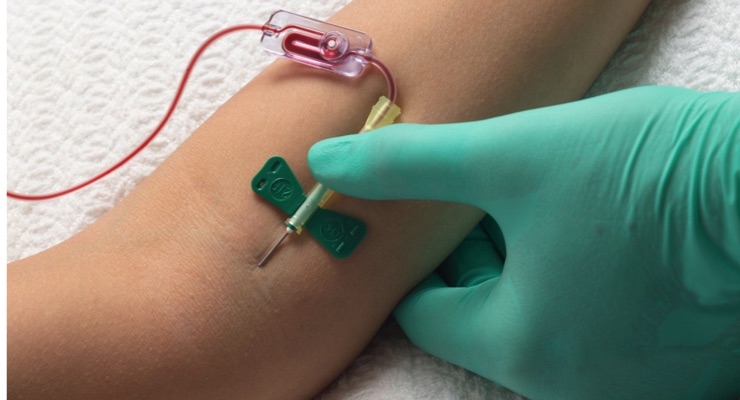
The product’s patented Kurin Lock automatically diverts approximately 0.15ml of initial blood and potential skin contaminants therein during blood culture collection. When skin microbes enter blood culture samples, false positive test results may misinform physicians and lead to unnecessary medical treatment, risk for patients, and significant costs for the hospital.
Data from the nine-month study was based on 4,200 blood cultures collected by phlebotomy and emergency department teams at Bayfront Health in St. Petersburg, Fla. During the trial, in which Kurin was employed for only 50 percent of all blood culture collections, the overall contamination rate inclusive of cultures taken both with and without Kurin, dropped by 54 percent from 2.5 percent to 1.2 percent, a statistically significant decline. When limiting the analysis to collections for which Kurin was used, there was only one contamination, which equates to approximately a 90 percent decrease in the contamination rate. The hospital estimates that each false positive blood culture adds $7,500 in costs, so implementing Kurin would save the hospital over $500,000 annually, after the cost of the product.
“This study focuses on total blood culture contaminants, and not just vacutainer collections. Without stratification, the mentioned results are what a user could expect to reproduce in a 'real world' scenario,” reported Jared Sutton, MPH, CIC, director of infection prevention at Bayfront Health, and his colleagues.
Kurin CEO Bob Rogers said, “As we have seen elsewhere, this study is additional confirmation that effective diversion of skin contaminants can be accomplished with approximately 0.15ml of blood waste and without requiring clinicians to change their standard practice. The accumulating data proves that Kurin can help hospitals prevent blood culture contamination, even in fast-paced EDs [emergency departments].”
Kurin Corporation, a certified minority business enterprise, is focused on the design, development, manufacture, marketing, and sale of products that help healthcare providers reduce contaminated blood cultures. Kurin, manufactured in San Diego, Calif., has received FDA 510(k) market clearance.
The product’s patented Kurin Lock automatically diverts approximately 0.15ml of initial blood and potential skin contaminants therein during blood culture collection. When skin microbes enter blood culture samples, false positive test results may misinform physicians and lead to unnecessary medical treatment, risk for patients, and significant costs for the hospital.
Data from the nine-month study was based on 4,200 blood cultures collected by phlebotomy and emergency department teams at Bayfront Health in St. Petersburg, Fla. During the trial, in which Kurin was employed for only 50 percent of all blood culture collections, the overall contamination rate inclusive of cultures taken both with and without Kurin, dropped by 54 percent from 2.5 percent to 1.2 percent, a statistically significant decline. When limiting the analysis to collections for which Kurin was used, there was only one contamination, which equates to approximately a 90 percent decrease in the contamination rate. The hospital estimates that each false positive blood culture adds $7,500 in costs, so implementing Kurin would save the hospital over $500,000 annually, after the cost of the product.
“This study focuses on total blood culture contaminants, and not just vacutainer collections. Without stratification, the mentioned results are what a user could expect to reproduce in a 'real world' scenario,” reported Jared Sutton, MPH, CIC, director of infection prevention at Bayfront Health, and his colleagues.
Kurin CEO Bob Rogers said, “As we have seen elsewhere, this study is additional confirmation that effective diversion of skin contaminants can be accomplished with approximately 0.15ml of blood waste and without requiring clinicians to change their standard practice. The accumulating data proves that Kurin can help hospitals prevent blood culture contamination, even in fast-paced EDs [emergency departments].”
Kurin Corporation, a certified minority business enterprise, is focused on the design, development, manufacture, marketing, and sale of products that help healthcare providers reduce contaminated blood cultures. Kurin, manufactured in San Diego, Calif., has received FDA 510(k) market clearance.