Business Wire07.12.18
JC Medical Inc. announced the treatment of the first patient with the company’s Transfemoral TAVI device, the J-Valve TF System for aortic regurgitation. The first patient was treated at St. Paul’s Hospital in Vancouver, Canada, by John Webb, MD, FACC, FSCAI, director of Interventional Cardiology.
There are currently no transcatheter valves approved for the treatment of pure aortic regurgitation in the United States, Canada, or Europe. Failing aortic valves make up a significant portion of patients with heart failure, a major public health problem with a prevalence of over 1.5 million in the United States and over 18 million worldwide.
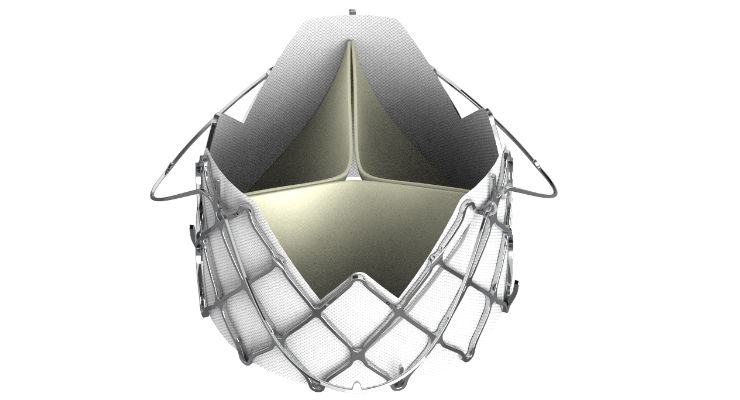
JC Medical’s J-Valve Systems are designed to restore normal blood flow out of the heart and into the body, which may improve symptoms of heart failure such as shortness of breath, fatigue and chest pain. The devices feature a proprietary anchor mechanism that does not require calcification for fixation and that is flexibly linked to a self-expanding stent frame to uniquely attach to the failing native heart valve.
“We are very pleased that our J-Valve could help this patient. We developed the J-Valve to be reliable and easy to use,” said Ji Zhang, M.D., founder and CEO of JC Medical. “We thank the physician group who has recognized this unique clinical solution. With approximately 2 percent of the elderly population having heart failure due to a defective stenotic aortic valve and 1 percent due to a regurgitant valve, this leads to a significant decrease in the quality of life, the loss of life for many, and costs the healthcare system billions of dollars annually. We look forward to further exploring the promise of J-Valve and its role in treating failing aortic heart valves.”
JC Medical Inc. is a private, venture-backed medical device company transforming the treatment of aortic valve disease, a growing clinical need in the face of the prevalence of heart disease due to a failing aortic valve and an aging population.
The J-Valve Transapical System received the China Food and Drug Administration approval in April 2017 and is currently available commercially in China. The J-Valve Transfemoral System is not available for sale in the United States, China, Europe, Canada or Japan.
There are currently no transcatheter valves approved for the treatment of pure aortic regurgitation in the United States, Canada, or Europe. Failing aortic valves make up a significant portion of patients with heart failure, a major public health problem with a prevalence of over 1.5 million in the United States and over 18 million worldwide.
JC Medical’s J-Valve Systems are designed to restore normal blood flow out of the heart and into the body, which may improve symptoms of heart failure such as shortness of breath, fatigue and chest pain. The devices feature a proprietary anchor mechanism that does not require calcification for fixation and that is flexibly linked to a self-expanding stent frame to uniquely attach to the failing native heart valve.
“We are very pleased that our J-Valve could help this patient. We developed the J-Valve to be reliable and easy to use,” said Ji Zhang, M.D., founder and CEO of JC Medical. “We thank the physician group who has recognized this unique clinical solution. With approximately 2 percent of the elderly population having heart failure due to a defective stenotic aortic valve and 1 percent due to a regurgitant valve, this leads to a significant decrease in the quality of life, the loss of life for many, and costs the healthcare system billions of dollars annually. We look forward to further exploring the promise of J-Valve and its role in treating failing aortic heart valves.”
JC Medical Inc. is a private, venture-backed medical device company transforming the treatment of aortic valve disease, a growing clinical need in the face of the prevalence of heart disease due to a failing aortic valve and an aging population.
The J-Valve Transapical System received the China Food and Drug Administration approval in April 2017 and is currently available commercially in China. The J-Valve Transfemoral System is not available for sale in the United States, China, Europe, Canada or Japan.