U.S. Food and Drug Administration07.02.18
The U.S. Food and Drug Administration today approved a new device, the Zephyr Endobronchial Valve (Zephyr Valve), intended to treat breathing difficulty associated with severe emphysema.
“Treatment options are limited for people with emphysema who have severe symptoms that have not improved from taking medicines. These have included lung surgery, such as lung volume reduction or lung transplants, which may not be suitable or appropriate for all patients,” said Tina Kiang, Ph.D., acting director, Division of Anesthesiology, General Hospital, Respiratory, Infection Control and Dental Devices, in the FDA’s Center for Devices and Radiological Health. “This novel device is a less invasive treatment that expands the options available to patients.”
The Centers for Disease Control and Prevention estimates that 3.5 million American adults have been diagnosed with emphysema. Emphysema, including severe emphysema, is a type of chronic obstructive pulmonary disease (COPD) due to damage to the air sacs (alveoli) in the lungs. Lung damage from emphysema is irreversible. The damaged alveoli can cause used air to become trapped in the lungs during exhalation. This can cause the diseased parts of the lung to get larger and put pressure on the healthy part of the lung, which makes it difficult to breathe. As a result, the body may not get the oxygen it needs.
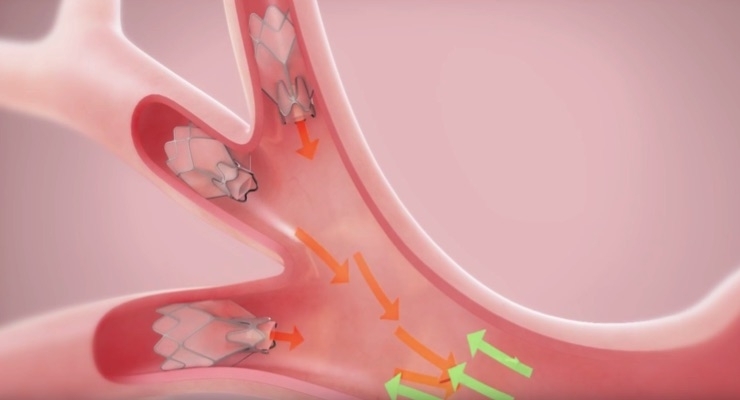
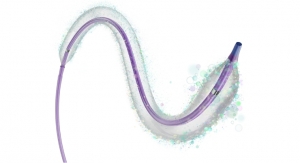
Using a flexible bronchoscope, a doctor places Zephyr Valves, similar in size to pencil erasers, into the diseased areas of the lung airways during a procedure in a hospital setting. Design of the device is intended to prevent air from entering the damaged parts of the lung and allow trapped air and fluids to escape. During inhalation, the valves close, preventing air from entering the damaged part of the lung and during exhalation, the valves open, letting out trapped air, which is intended to relieve pressure.
The FDA reviewed data from a multi-center study of 190 patients with severe emphysema. In this study, 128 patients were treated with Zephyr Valves and medical management according to current clinical guidelines, including medications (bronchodilators, corticosteroids, antibiotics or anti-inflammatory maintenance medications) and pulmonary rehabilitation, while 62 patients (the control group) received medical management only. Results of treatment were measured by how many patients in each arm of the study had at least a 15 percent improvement in pulmonary function scores (the volume of air that can forcibly be blown out in one second after full inhalation). At one year, 47.7 percent of patients treated with Zephyr Valves experienced at least a 15 percent improvement in their pulmonary function scores, compared with 16.8 percent of patients in the control group. Adverse events observed in the study include death, air leak (pneumothorax), pneumonia, worsening of emphysema, coughing up blood, shortness of breath and chest pain.
The Zephyr Valve device is contraindicated for patients with active lung infections; those who are allergic to nitinol, nickel, titanium or silicone; active smokers and those who are not able to tolerate the bronchoscopic procedure. Patients who have had major lung procedures, heart disease, large bubbles of air trapped in the lung or who have not responded to other treatments should talk with their providers to determine if the Zephyr Valve device is appropriate for them.
The Zephyr Valve was granted Breakthrough Device designation, meaning the FDA provided intensive interaction and guidance to the company on efficient device development, to expedite evidence generation and the agency’s review of the device. To qualify for such designation, a device must provide for more effective treatment or diagnosis of a life-threatening or irreversibly debilitating disease or condition, and meet one of the following criteria: the device must represent a breakthrough technology; there must be no approved or cleared alternatives; the device must offer significant advantages over existing approved or cleared alternatives; or the availability of the device is in the best interest of patients.
The FDA reviewed the Zephyr Valve device through the premarket approval review pathway, a regulatory pathway for the highest risk class of devices.
The FDA granted approval of the Zephyr Valve device to Pulmonx Inc.
“Treatment options are limited for people with emphysema who have severe symptoms that have not improved from taking medicines. These have included lung surgery, such as lung volume reduction or lung transplants, which may not be suitable or appropriate for all patients,” said Tina Kiang, Ph.D., acting director, Division of Anesthesiology, General Hospital, Respiratory, Infection Control and Dental Devices, in the FDA’s Center for Devices and Radiological Health. “This novel device is a less invasive treatment that expands the options available to patients.”
The Centers for Disease Control and Prevention estimates that 3.5 million American adults have been diagnosed with emphysema. Emphysema, including severe emphysema, is a type of chronic obstructive pulmonary disease (COPD) due to damage to the air sacs (alveoli) in the lungs. Lung damage from emphysema is irreversible. The damaged alveoli can cause used air to become trapped in the lungs during exhalation. This can cause the diseased parts of the lung to get larger and put pressure on the healthy part of the lung, which makes it difficult to breathe. As a result, the body may not get the oxygen it needs.
Using a flexible bronchoscope, a doctor places Zephyr Valves, similar in size to pencil erasers, into the diseased areas of the lung airways during a procedure in a hospital setting. Design of the device is intended to prevent air from entering the damaged parts of the lung and allow trapped air and fluids to escape. During inhalation, the valves close, preventing air from entering the damaged part of the lung and during exhalation, the valves open, letting out trapped air, which is intended to relieve pressure.
The FDA reviewed data from a multi-center study of 190 patients with severe emphysema. In this study, 128 patients were treated with Zephyr Valves and medical management according to current clinical guidelines, including medications (bronchodilators, corticosteroids, antibiotics or anti-inflammatory maintenance medications) and pulmonary rehabilitation, while 62 patients (the control group) received medical management only. Results of treatment were measured by how many patients in each arm of the study had at least a 15 percent improvement in pulmonary function scores (the volume of air that can forcibly be blown out in one second after full inhalation). At one year, 47.7 percent of patients treated with Zephyr Valves experienced at least a 15 percent improvement in their pulmonary function scores, compared with 16.8 percent of patients in the control group. Adverse events observed in the study include death, air leak (pneumothorax), pneumonia, worsening of emphysema, coughing up blood, shortness of breath and chest pain.
The Zephyr Valve device is contraindicated for patients with active lung infections; those who are allergic to nitinol, nickel, titanium or silicone; active smokers and those who are not able to tolerate the bronchoscopic procedure. Patients who have had major lung procedures, heart disease, large bubbles of air trapped in the lung or who have not responded to other treatments should talk with their providers to determine if the Zephyr Valve device is appropriate for them.
The Zephyr Valve was granted Breakthrough Device designation, meaning the FDA provided intensive interaction and guidance to the company on efficient device development, to expedite evidence generation and the agency’s review of the device. To qualify for such designation, a device must provide for more effective treatment or diagnosis of a life-threatening or irreversibly debilitating disease or condition, and meet one of the following criteria: the device must represent a breakthrough technology; there must be no approved or cleared alternatives; the device must offer significant advantages over existing approved or cleared alternatives; or the availability of the device is in the best interest of patients.
The FDA reviewed the Zephyr Valve device through the premarket approval review pathway, a regulatory pathway for the highest risk class of devices.
The FDA granted approval of the Zephyr Valve device to Pulmonx Inc.