PR Newswire04.23.18
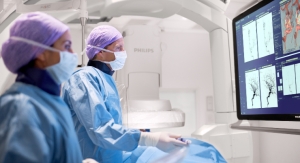
Viz.ai Inc., an applied artificial intelligence healthcare company, today announced that the U.S. Food and Drug Administration (FDA) has cleared Viz CTP through the 510(k) pathway. Viz CTP offers image processing software to view and analyze functional and dynamic CT perfusion images. This clearance offers healthcare providers another important tool for automated cerebral-image analysis.
With the addition of Viz CTP, Viz.ai now offers perfusion analysis to complement a comprehensive suite of technology, which provides automated large vessel occlusion (LVO) stroke identification, stroke triage and LVO notification, patient selection, HIPAA compliant communication, mobile medical image viewing and transport coordination.
"In stroke, good patient outcomes can come down to a matter of minutes," said Dr. Chris Mansi, neurosurgeon, CEO and co-founder of Viz.ai Inc. "We are proud to offer hospital systems a comprehensive solution that can facilitate end-to-end care coordination and create efficiencies in acute stroke pathways to help get the right patient to the right doctor at the right time."
With the addition of Viz CTP, Viz.ai now offers perfusion analysis to complement a comprehensive suite of technology, which provides automated large vessel occlusion (LVO) stroke identification, stroke triage and LVO notification, patient selection, HIPAA compliant communication, mobile medical image viewing and transport coordination.
"In stroke, good patient outcomes can come down to a matter of minutes," said Dr. Chris Mansi, neurosurgeon, CEO and co-founder of Viz.ai Inc. "We are proud to offer hospital systems a comprehensive solution that can facilitate end-to-end care coordination and create efficiencies in acute stroke pathways to help get the right patient to the right doctor at the right time."