Business Wire03.16.18
RenovaCare Inc., developer of the SkinGun and CellMist System for spraying a patient’s own skin stem cells onto burns and wounds for rapid self-healing, has won against patent challenger Avita Medical in a proceeding at the United States Patent Office (USPTO).
Noting that Avita’s arguments were based on old claims and technologies which had already been considered and rejected, the USPTO dismissed the challenge and denied a trial sought by Avita.
“I’m proud of the technologies developed by our team for the millions that suffer from burns and wounds and deserve access to the very latest in medical innovation,” stated Thomas Bold, president and CEO of RenovaCare. “Just as we’ve done with this challenge, we’ll aggressively protect our intellectual property while continuing to expand our patent portfolio and enhance shareholder value.”
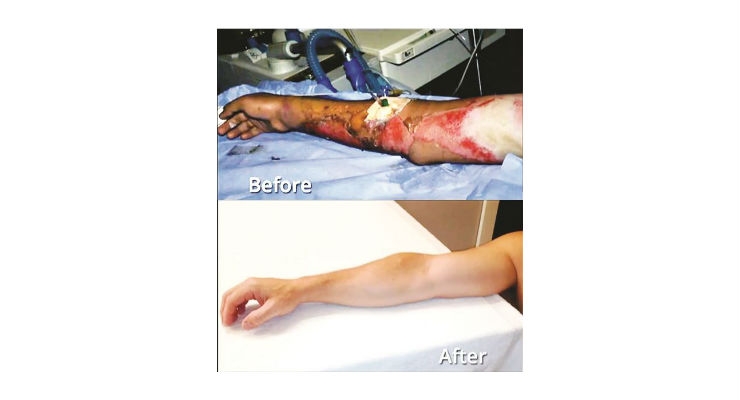
To-date more than 70 human patients with severe second-degree burns have been treated with the technology behind the RenovaCare SkinGun and its patents. Clinical results, case studies and data have been presented in numerous peer-reviewed publications.
Treated with a gentle spray of their own skin stem cells, many patients have left the hospital within a matter of days, healed with full mobility and scar-free.
In contrast, patients who undergo skin grafting, today’s default treatment, can remain hospitalized for weeks and months while enduring multiple painful surgeries and prolonged physical therapy. Skin graft patients can suffer with the painful psychological effects of disfigurement caused by permanent scarring and are often forced to cope with the ongoing use of pain medications and protracted joint mobility issues.
Another option, in-vitro cultured epithelial grafts, requires harvesting cells from a patient, which are then transported to a specialized external laboratory where they take weeks to form sheets of skin. These fragile sheets must then be shipped back to the hospital for surgical stitching onto a patient’s wounds, a process that is complicated, time-consuming and expensive.
RenovaCare has developed its novel SkinGun for the ultra-gentle spray of a patient’s own skin stem cells on to burns and wounds for rapid healing. The potent, regenerative cells are isolated from a postage stamp sized donor site and suspended in a liquid, the CellMist Solution. The CellMist Solution is then sprayed on to wounds using the SkinGun. The entire procedure can be completed in as little as 90 minutes.
The SkinGun spray device and cell isolation methodology are subjects of patents awarded to RenovaCare in the United States and Europe. The company has also filed additional patent applications and continues expanding its intellectual property portfolio.
*RenovaCare products are currently in development. They are not available for sale in the United States.
RenovaCare Inc. is developing first-of-its-kind autologous (self-donated) stem cell therapies for the regeneration of human organs, and novel medical grade liquid sprayer devices. Its initial product under development targets the body’s largest organ, the skin. The company’s flagship technology, the CellMist System, uses its patented SkinGun to spray a liquid suspension of a patient’s stem cells (the CellMist Solution) onto wounds. RenovaCare is developing its CellMist System as a new alternative for patients suffering from burns, chronic and acute wounds, and scars. In the United States alone, this $45 billion market is greater than the spending on high-blood pressure management, cholesterol treatments, and back pain therapeutics.
Noting that Avita’s arguments were based on old claims and technologies which had already been considered and rejected, the USPTO dismissed the challenge and denied a trial sought by Avita.
“I’m proud of the technologies developed by our team for the millions that suffer from burns and wounds and deserve access to the very latest in medical innovation,” stated Thomas Bold, president and CEO of RenovaCare. “Just as we’ve done with this challenge, we’ll aggressively protect our intellectual property while continuing to expand our patent portfolio and enhance shareholder value.”
To-date more than 70 human patients with severe second-degree burns have been treated with the technology behind the RenovaCare SkinGun and its patents. Clinical results, case studies and data have been presented in numerous peer-reviewed publications.
Treated with a gentle spray of their own skin stem cells, many patients have left the hospital within a matter of days, healed with full mobility and scar-free.
In contrast, patients who undergo skin grafting, today’s default treatment, can remain hospitalized for weeks and months while enduring multiple painful surgeries and prolonged physical therapy. Skin graft patients can suffer with the painful psychological effects of disfigurement caused by permanent scarring and are often forced to cope with the ongoing use of pain medications and protracted joint mobility issues.
Another option, in-vitro cultured epithelial grafts, requires harvesting cells from a patient, which are then transported to a specialized external laboratory where they take weeks to form sheets of skin. These fragile sheets must then be shipped back to the hospital for surgical stitching onto a patient’s wounds, a process that is complicated, time-consuming and expensive.
RenovaCare has developed its novel SkinGun for the ultra-gentle spray of a patient’s own skin stem cells on to burns and wounds for rapid healing. The potent, regenerative cells are isolated from a postage stamp sized donor site and suspended in a liquid, the CellMist Solution. The CellMist Solution is then sprayed on to wounds using the SkinGun. The entire procedure can be completed in as little as 90 minutes.
The SkinGun spray device and cell isolation methodology are subjects of patents awarded to RenovaCare in the United States and Europe. The company has also filed additional patent applications and continues expanding its intellectual property portfolio.
*RenovaCare products are currently in development. They are not available for sale in the United States.
RenovaCare Inc. is developing first-of-its-kind autologous (self-donated) stem cell therapies for the regeneration of human organs, and novel medical grade liquid sprayer devices. Its initial product under development targets the body’s largest organ, the skin. The company’s flagship technology, the CellMist System, uses its patented SkinGun to spray a liquid suspension of a patient’s stem cells (the CellMist Solution) onto wounds. RenovaCare is developing its CellMist System as a new alternative for patients suffering from burns, chronic and acute wounds, and scars. In the United States alone, this $45 billion market is greater than the spending on high-blood pressure management, cholesterol treatments, and back pain therapeutics.