U.S. Food and Drug Administration02.26.18
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death.
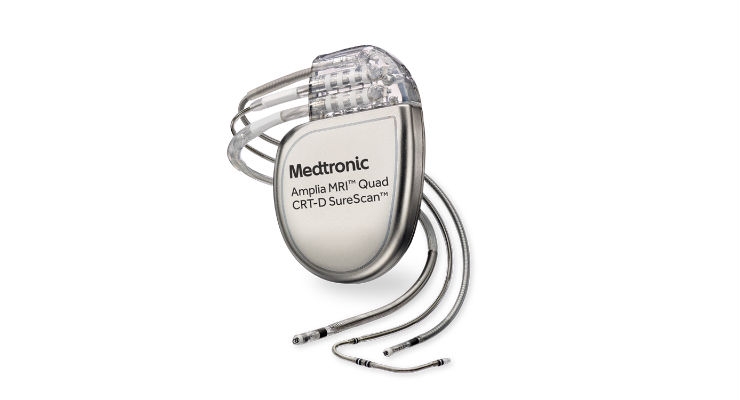
Implantable Cardioverter Defibrillators (ICDs) and Cardiac Resynchronization Therapy Defibrillators (CRT-Ds) are devices that provide pacing for slow heart rhythms, and electrical shock or pacing to stop dangerously fast heart rhythms.
ICDs and CRT-Ds are both implanted under the skin in the upper chest area with connecting insulated wires called "leads" that go into the heart. Patients need an ICD or CRT-D if their heartbeat is too slow (bradycardia), too fast (tachycardia), or needs coordination to treat heart failure.
Medtronic is recalling certain ICDs and CRT-Ds due to a defect in the manufacturing process. This defect causes an out of specification gas mixture inside the device and may prevent the device from delivering the electrical shock needed to pace a patient’s heartbeat or revive a patient in cardiac arrest.
The delay or inability to deliver a shock to a patient in cardiac arrest or pace a patient’s heart whose heartbeat is too slow could result in serious injury and/or death.
Who Is Affected?
What to Do:
On January 22, 2018 Medtronic sent an Urgent Medical Device Recall notice to affected customers. The notice asked customers to:
Medtronic will offer a supplemental device warranty for affected devices.
- Recalled Product(s): Medtronic Cardiac Resynchronization Therapy with Defibrillation (CRT-Ds) and Implantable Cardiovert-Defibrillators (ICDs)
- Product Codes: NIK, LWS
- Serial Numbers: See Complete List
- Manufacturing Dates: July 13, 2013 to August 8, 2017
- Devices Recalled in the U.S.: 48 units nationwide
Implantable Cardioverter Defibrillators (ICDs) and Cardiac Resynchronization Therapy Defibrillators (CRT-Ds) are devices that provide pacing for slow heart rhythms, and electrical shock or pacing to stop dangerously fast heart rhythms.
ICDs and CRT-Ds are both implanted under the skin in the upper chest area with connecting insulated wires called "leads" that go into the heart. Patients need an ICD or CRT-D if their heartbeat is too slow (bradycardia), too fast (tachycardia), or needs coordination to treat heart failure.
Medtronic is recalling certain ICDs and CRT-Ds due to a defect in the manufacturing process. This defect causes an out of specification gas mixture inside the device and may prevent the device from delivering the electrical shock needed to pace a patient’s heartbeat or revive a patient in cardiac arrest.
The delay or inability to deliver a shock to a patient in cardiac arrest or pace a patient’s heart whose heartbeat is too slow could result in serious injury and/or death.
Who Is Affected?
- Patients with an affected Medtronic ICD or CRT-D device
- Caregivers of patients with an affected Medtronic ICD or CRT-D device
- Healthcare providers treating patients with heart failure or heart rhythm problems using affected Medtronic ICD or CRT-D devices
What to Do:
On January 22, 2018 Medtronic sent an Urgent Medical Device Recall notice to affected customers. The notice asked customers to:
- Consider Prophylactic device replacement for patients who have been implanted with one of the affected devices.
- Contact their Medtronic sales representative for terms and conditions for device warranties.
- Review the recall notice and ensure appropriate staff is aware of the notice.
Medtronic will offer a supplemental device warranty for affected devices.