Business Wire01.29.18
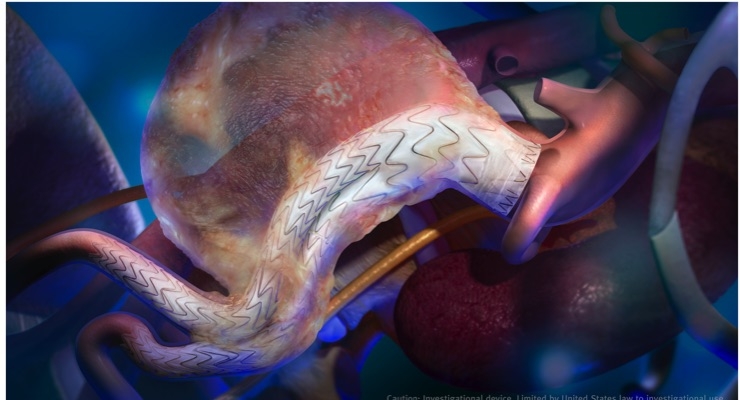
W. L. Gore & Associates Inc. (Gore) has announced the first implant of the GORE EXCLUDER Conformable AAA Endoprosthesis in the United States. The successful procedure took place on Dec. 19, 2017, at Maimonides Medical Center in New York, N.Y., by Robert Rhee, M.D., chief of Vascular and Endovascular Surgery, and national principal investigator.
This EVAR device, which is the first to feature angulation control, is part of an investigational clinical study approved by the U.S. Food and Drug Administration (FDA). The clinical study will assess the safety and effectiveness of the device in treating infrarenal abdominal aortic aneurysms (AAA) in patients with challenging anatomy. The clinical study consists of two sub-studies, each assessing the device for a different range of patient anatomies. The implantation by Rhee is part of the short neck sub-study to assess the device in aortic neck angles of 0 degrees to 60 degrees and aortic neck lengths of 10 mm or greater. The high neck angulation sub-study will evaluate proximal aortic neck angles of 61 degrees to 90 degrees and aortic neck lengths of 10 mm or greater.
“Patients with extreme proximal neck anatomies often do not qualify for EVAR. Regardless of device flexibility, current delivery systems make it difficult to achieve conformability during deployment,” said Rhee. “The angulation control in the new delivery system for the GORE EXCLUDER Conformable AAA Device is intended to allow physicians a greater level of control to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen.”
The GORE EXCLUDER Conformable AAA Device builds on the proven clinical performance of the GORE EXCLUDER Device, which represents 20 years of worldwide experience and more than 300,000 patients treated,* a history unmatched by currently available AAA stent grafts. The new device leverages the limb design of the GORE EXCLUDER Device which has demonstrated clinical performance as evidenced by 0.5 percent limb occlusion through three-year follow-up.** The limbs, a unique combination of proprietary ePTFE graft material and a fully supported, nested, nitinol stent, are designed to prevent kinking and occlusion.
The GORE EXCLUDER Conformable AAA Device debuts the GORE ACTIVE CONTROL System into the abdominal aorta. This delivery system includes angulation control, giving physicians the option to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen and to maximize the conformability and seal of the device. Like the GORE C3 Delivery System, the GORE ACTIVE CONTROL System offers the ability to reposition the device if needed after initial deployment to achieve optimal device placement.
“This implant is the latest step in our continuing efforts to offer the broadest and most technically advanced endovascular treatment capabilities on the market,” said Eric Zacharias, Vascular Business leader. “Since bringing our first EVAR device to market two decades ago, Gore has been committed to providing innovative solutions that expand the endovascular treatment of aortic disease. In collaborating with our physician partners, we heard that highly angulated anatomy can challenge the ability of currently available EVAR devices to achieve an optimal seal. The IDE study will evaluate the ability of the system to fill the unmet need for an EVAR device to treat that anatomy. The system combines the nesting stent design proven successful in the Conformable GORE TAG Thoracic Endoprosthesis with the repositionability of the GORE C3 Delivery System, while the new angulation control feature meets the need for an EVAR device that can treat more challenging anatomies."
The GORE EXCLUDER Conformable AAA Device is part of the family of endovascular products that share a mission to effectively treat aortic disease, backed by Gore’s clinical support team and educational offerings.† The comprehensive portfolio of products includes the Conformable GORE TAG Thoracic Endoprosthesis for the treatment of thoracic aneurysms, transections, and Type B dissections; the GORE EXCLUDER Iliac Branch Endoprosthesis (IBE), the first FDA-approved off-the-shelf device indicated for the endovascular treatment of common iliac artery aneurysms or aortoiliac aneurysms; the GORE VIABAHN Endoprosthesis, the stent graft for the treatment of complex peripheral vascular disease; and the GORE VIABAHN VBX Balloon Expandable Endoprosthesis, the first FDA-approved balloon-expandable stent graft for treatment of de novo or restenotic lesions found in iliac arteries, including lesions at the aortic bifurcation.
For potential additions to Gore’s branched portfolio, investigational studies are ongoing for the GORE EXCLUDER Thoracoabdominal Branch Endoprosthesis (TAMBE), and enrollment continues in the GORE TAG Thoracic Branch Endoprosthesis (TBE) Pivotal Study to assess safety and effectiveness in treating lesions of the aortic arch and descending thoracic aorta, while maintaining flow into a single aortic arch branch vessel. Both of these are investigational devices that are not yet approved for commercial sale.
* Based on the number of Trunk-Ipsilateral Legs distributed.
·
** GREAT. n = 3,273. To calculate the overall event rates from procedure through end of study period, all subjects who could have had events, regardless of length of follow-up, were included. For outcome data, GREAT only collects site reported serious adverse events.
† For complete indications and other important safety information for Gore commercial products referenced herein, refer to the applicable Instructions for Use (IFU).
Gore Medical Products Division engineers devices that treat a range of cardiovascular and other health conditions. With more than 40 million medical devices implanted over the course of more than 40 years, Gore builds on its legacy of improving patient outcomes through research, education and quality initiatives. Product performance, ease of use and quality of service provide sustainable cost savings for physicians, hospitals and insurers.
W. L. Gore & Associates is a global materials science company dedicated to transforming industries and improving lives. Founded in 1958, Gore has built a reputation for solving complex technical challenges in the most demanding environments—from revolutionizing the outerwear industry with GORE-TEX fabric to creating medical devices that improve and save lives to enabling new levels of performance in the aerospace, pharmaceutical and mobile electronics markets, among other industries. Headquartered in Newark, Del., Gore employs approximately 9,500 associates and generates annual revenues that exceed $3 billion.
This EVAR device, which is the first to feature angulation control, is part of an investigational clinical study approved by the U.S. Food and Drug Administration (FDA). The clinical study will assess the safety and effectiveness of the device in treating infrarenal abdominal aortic aneurysms (AAA) in patients with challenging anatomy. The clinical study consists of two sub-studies, each assessing the device for a different range of patient anatomies. The implantation by Rhee is part of the short neck sub-study to assess the device in aortic neck angles of 0 degrees to 60 degrees and aortic neck lengths of 10 mm or greater. The high neck angulation sub-study will evaluate proximal aortic neck angles of 61 degrees to 90 degrees and aortic neck lengths of 10 mm or greater.
“Patients with extreme proximal neck anatomies often do not qualify for EVAR. Regardless of device flexibility, current delivery systems make it difficult to achieve conformability during deployment,” said Rhee. “The angulation control in the new delivery system for the GORE EXCLUDER Conformable AAA Device is intended to allow physicians a greater level of control to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen.”
The GORE EXCLUDER Conformable AAA Device builds on the proven clinical performance of the GORE EXCLUDER Device, which represents 20 years of worldwide experience and more than 300,000 patients treated,* a history unmatched by currently available AAA stent grafts. The new device leverages the limb design of the GORE EXCLUDER Device which has demonstrated clinical performance as evidenced by 0.5 percent limb occlusion through three-year follow-up.** The limbs, a unique combination of proprietary ePTFE graft material and a fully supported, nested, nitinol stent, are designed to prevent kinking and occlusion.
The GORE EXCLUDER Conformable AAA Device debuts the GORE ACTIVE CONTROL System into the abdominal aorta. This delivery system includes angulation control, giving physicians the option to angle or bend the device to achieve orthogonal placement to the aortic blood flow lumen and to maximize the conformability and seal of the device. Like the GORE C3 Delivery System, the GORE ACTIVE CONTROL System offers the ability to reposition the device if needed after initial deployment to achieve optimal device placement.
“This implant is the latest step in our continuing efforts to offer the broadest and most technically advanced endovascular treatment capabilities on the market,” said Eric Zacharias, Vascular Business leader. “Since bringing our first EVAR device to market two decades ago, Gore has been committed to providing innovative solutions that expand the endovascular treatment of aortic disease. In collaborating with our physician partners, we heard that highly angulated anatomy can challenge the ability of currently available EVAR devices to achieve an optimal seal. The IDE study will evaluate the ability of the system to fill the unmet need for an EVAR device to treat that anatomy. The system combines the nesting stent design proven successful in the Conformable GORE TAG Thoracic Endoprosthesis with the repositionability of the GORE C3 Delivery System, while the new angulation control feature meets the need for an EVAR device that can treat more challenging anatomies."
The GORE EXCLUDER Conformable AAA Device is part of the family of endovascular products that share a mission to effectively treat aortic disease, backed by Gore’s clinical support team and educational offerings.† The comprehensive portfolio of products includes the Conformable GORE TAG Thoracic Endoprosthesis for the treatment of thoracic aneurysms, transections, and Type B dissections; the GORE EXCLUDER Iliac Branch Endoprosthesis (IBE), the first FDA-approved off-the-shelf device indicated for the endovascular treatment of common iliac artery aneurysms or aortoiliac aneurysms; the GORE VIABAHN Endoprosthesis, the stent graft for the treatment of complex peripheral vascular disease; and the GORE VIABAHN VBX Balloon Expandable Endoprosthesis, the first FDA-approved balloon-expandable stent graft for treatment of de novo or restenotic lesions found in iliac arteries, including lesions at the aortic bifurcation.
For potential additions to Gore’s branched portfolio, investigational studies are ongoing for the GORE EXCLUDER Thoracoabdominal Branch Endoprosthesis (TAMBE), and enrollment continues in the GORE TAG Thoracic Branch Endoprosthesis (TBE) Pivotal Study to assess safety and effectiveness in treating lesions of the aortic arch and descending thoracic aorta, while maintaining flow into a single aortic arch branch vessel. Both of these are investigational devices that are not yet approved for commercial sale.
* Based on the number of Trunk-Ipsilateral Legs distributed.
·
** GREAT. n = 3,273. To calculate the overall event rates from procedure through end of study period, all subjects who could have had events, regardless of length of follow-up, were included. For outcome data, GREAT only collects site reported serious adverse events.
† For complete indications and other important safety information for Gore commercial products referenced herein, refer to the applicable Instructions for Use (IFU).
Gore Medical Products Division engineers devices that treat a range of cardiovascular and other health conditions. With more than 40 million medical devices implanted over the course of more than 40 years, Gore builds on its legacy of improving patient outcomes through research, education and quality initiatives. Product performance, ease of use and quality of service provide sustainable cost savings for physicians, hospitals and insurers.
W. L. Gore & Associates is a global materials science company dedicated to transforming industries and improving lives. Founded in 1958, Gore has built a reputation for solving complex technical challenges in the most demanding environments—from revolutionizing the outerwear industry with GORE-TEX fabric to creating medical devices that improve and save lives to enabling new levels of performance in the aerospace, pharmaceutical and mobile electronics markets, among other industries. Headquartered in Newark, Del., Gore employs approximately 9,500 associates and generates annual revenues that exceed $3 billion.