GlobeNewswire01.16.18
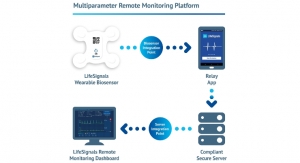
Biotricity Inc., a medical diagnostic and consumer healthcare technology company dedicated to delivering biometric remote monitoring solutions, announced it has commenced extending the capabilities of its remote patient monitoring (RPM) platform with artificial intelligence (AI) to differentiate itself within the growing remote monitoring marketplace. The company is already working with a proof of concept version of its RPM hardware with embedded AI. Biotricity intends the first commercial application to be the next generation of its Bioflux device. The company aims to file an additional 510(k) hardware clearance with the U.S. Food and Drug Administration (FDA) by Q3 in 2018, before launching the next version. With Bioflux’s recent FDA clearance, development of AI and next generation hardware will occur while the current version of Bioflux is in the marketplace.
“We plan on implementing artificial intelligence at the device level to maximize detection and accuracy while increasing efficiency,” said Waqaas Al-Siddiq, Founder and CEO of Biotricity. “We expect our next generation Bioflux device with artificial intelligence capabilities to be the first of its kind in the RPM marketplace.”
Remote patient monitoring is a multi-billion dollar market that is rapidly growing as technology becomes smaller and connectivity cheaper. Biotricity intends to leverage its proprietary platform to continuously innovate within the RPM market.
“We plan on implementing artificial intelligence at the device level to maximize detection and accuracy while increasing efficiency,” said Waqaas Al-Siddiq, Founder and CEO of Biotricity. “We expect our next generation Bioflux device with artificial intelligence capabilities to be the first of its kind in the RPM marketplace.”
Remote patient monitoring is a multi-billion dollar market that is rapidly growing as technology becomes smaller and connectivity cheaper. Biotricity intends to leverage its proprietary platform to continuously innovate within the RPM market.