PR Newswire11.08.17
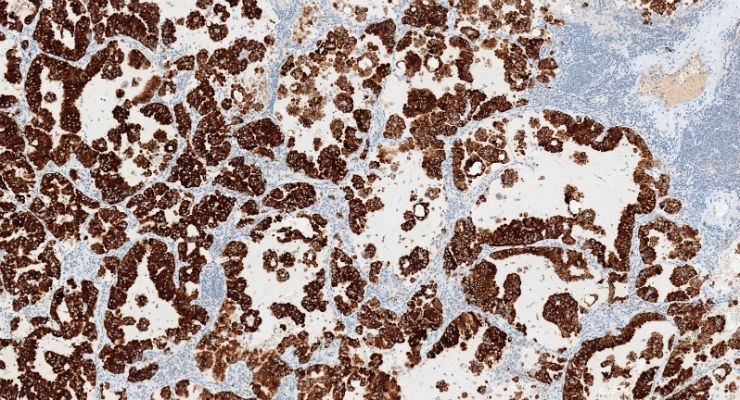
Roche announced it has obtained U.S. Food and Drug Administration (FDA) approval for the VENTANA ALK (D5F3) CDx Assay as a companion diagnostic to identify ALK-positive non-small cell lung cancer (NSCLC) patients eligible for treatment with the Roche medicine ALECENSA (alectinib). The VENTANA ALK (D5F3) CDx Assay is the only test FDA-approved as a companion diagnostic for ALECENSA.
"We are very excited about the companion diagnostic label expansion for the ALK (D5F3) assay. This immunohistochemistry assay will deliver critical information on treatment options for non-small cell lung cancer patients," said Ann Costello, head of Roche Tissue Diagnostics. “The ability to identify ALK-positive patients very quickly with a simple IHC test ensures that more patients can benefit from targeted therapy.”
The VENTANA ALK (D5F3) CDx Assay has been shown in studies to identify more patients that will benefit from an anti-ALK target therapy than fluorescent in situ hybridization (FISH) testing.1,2,3,4 The VENTANA ALK (D5F3) CDx Assay is available for use on the Roche BenchMark XT or BenchMark ULTRA IHC/ISH slide staining systems.5
References
1van der Wekken AJ, Pelgrim R, 't Hart N, Werner N, Mastik MF, et al. Dichotomous. ALK-IHC is a better predictor for ALK inhibition outcome than traditional ALK-FISH in advanced non-small cell lung cancer. Clinical Cancer Research, 2017, 23(15):4251-4258. doi: 10.1158/1078-0432.CCR-16-1631.
2Zhou J, Zhao J, Sun K, Wang B, Wang L, et al. Accurate and economical detection of ALK positive lung Adenocarcinoma with semiquantitative immunohistochemical screening. PLoS ONE 2014, 9(3): e92828.doi:10.1371/journal.pone.0092828.
3Shan L, Lian F, Guo L, Yang X, Ying J, et al. Combination of conventional immunohistochemistry and qRT-PCR to detect ALK rearrangement. Diagnostic Pathology 2014, 9:3.doi:10.1186/1746-1596-9-3.
4Ying J, Guo L, Qiu T, Shan L, Ling Y, et al. Diagnostic value of a novel fully automated immunochemistry assay for detection of ALK rearrangement in primary lung adenocarcinoma. Annals of Oncology, 2013, 24(10):2589-2593; doi:10.1093/annonc/mdt295.
5The approved ALK (D5F3) Assay stained with OptiView DAB Detection and Amp is a 4 1/2-hour, fully automated test to be stained with all other routine IHC testing for same-day results and to meet current CAP/IASLC/AMP guidelines for testing patients with lung cancer; Lindeman NI, Cagle PT, Beasley MB, Chitale DA, Dacic S, et al. Molecular testing guideline for selection of lung cancer patients for EGFR and ALK tyrosine kinase inhibitors: guideline from the College of American Pathologists, International Association for the Study of Lung Cancer, and Association for Molecular Pathology. Journal of Thoracic Oncology 2013; 8(7):823–859. doi: 10.1097/JTO.0b013e318290868f.
"We are very excited about the companion diagnostic label expansion for the ALK (D5F3) assay. This immunohistochemistry assay will deliver critical information on treatment options for non-small cell lung cancer patients," said Ann Costello, head of Roche Tissue Diagnostics. “The ability to identify ALK-positive patients very quickly with a simple IHC test ensures that more patients can benefit from targeted therapy.”
The VENTANA ALK (D5F3) CDx Assay has been shown in studies to identify more patients that will benefit from an anti-ALK target therapy than fluorescent in situ hybridization (FISH) testing.1,2,3,4 The VENTANA ALK (D5F3) CDx Assay is available for use on the Roche BenchMark XT or BenchMark ULTRA IHC/ISH slide staining systems.5
References
1van der Wekken AJ, Pelgrim R, 't Hart N, Werner N, Mastik MF, et al. Dichotomous. ALK-IHC is a better predictor for ALK inhibition outcome than traditional ALK-FISH in advanced non-small cell lung cancer. Clinical Cancer Research, 2017, 23(15):4251-4258. doi: 10.1158/1078-0432.CCR-16-1631.
2Zhou J, Zhao J, Sun K, Wang B, Wang L, et al. Accurate and economical detection of ALK positive lung Adenocarcinoma with semiquantitative immunohistochemical screening. PLoS ONE 2014, 9(3): e92828.doi:10.1371/journal.pone.0092828.
3Shan L, Lian F, Guo L, Yang X, Ying J, et al. Combination of conventional immunohistochemistry and qRT-PCR to detect ALK rearrangement. Diagnostic Pathology 2014, 9:3.doi:10.1186/1746-1596-9-3.
4Ying J, Guo L, Qiu T, Shan L, Ling Y, et al. Diagnostic value of a novel fully automated immunochemistry assay for detection of ALK rearrangement in primary lung adenocarcinoma. Annals of Oncology, 2013, 24(10):2589-2593; doi:10.1093/annonc/mdt295.
5The approved ALK (D5F3) Assay stained with OptiView DAB Detection and Amp is a 4 1/2-hour, fully automated test to be stained with all other routine IHC testing for same-day results and to meet current CAP/IASLC/AMP guidelines for testing patients with lung cancer; Lindeman NI, Cagle PT, Beasley MB, Chitale DA, Dacic S, et al. Molecular testing guideline for selection of lung cancer patients for EGFR and ALK tyrosine kinase inhibitors: guideline from the College of American Pathologists, International Association for the Study of Lung Cancer, and Association for Molecular Pathology. Journal of Thoracic Oncology 2013; 8(7):823–859. doi: 10.1097/JTO.0b013e318290868f.