Globe Newswire11.02.17
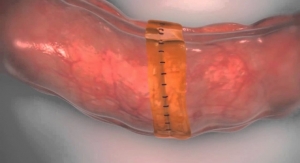
Painful post-surgical drains may become unnecessary for breast cancer patients who undergo a mastectomy procedure (removal of the breast). A level I clinical study seeks to provide evidence that the skin flap can be adhered to the chest wall with TissuGlu Surgical Adhesive after removal of the breast tissue, assisting the healing process and eliminating the need for drains. A drain-free recovery could help to make the experience easier for both mastectomy patients and their care providers.
The European study, with four centers in Germany and seven in the United Kingdom, started in February and has completed enrollment of nearly 80 patients, with half of the patients treated with post-surgical drains and the other half treated with TissuGlu and no drains. After a 90-day follow up period, the study will assess the number of clinical interventions related to wound healing and fluid management in both groups. Preliminary assessments indicate that the outcomes of the study are positive.
“Our patients have been very enthusiastic about participating in the study when they hear about the chance of a drain-free procedure,” said Polly King, principal investigator at the Royal Cornwall hospital in the United Kingdom, which recruited the highest number of patients for the study. “Patients without drains feel more comfortable about going home sooner than those who wake up from surgery with a drain.”
“TissuGlu has been used for flap fixation after mastectomy and several other ‘flap procedures’ in Europe for a few years now. The body of evidence required to bring this new approach into the mainstream has been steadily growing, and this multinational prospective randomized study really represents the ‘Gold Standard’ of clinical evidence,” said Mart Pearson, vice president of Europe for Cohera Medical Inc. “We hope that these results will open the door for many other breast cancer patients who would greatly prefer a recovery without drains.”
This study adds to the growing body of evidence of positive TissuGlu outcomes.
Cohera Medical Inc. is developing and commercializing a line of surgical adhesives and sealants. Cohera Medical’s products are based on a chemical design that is resorbable, non-toxic, and easy-to-use. The company’s lead product, TissuGlu Surgical Adhesive, is indicated for use in the United States for the approximation of tissue planes in abdominoplasty procedures. TissuGlu is currently approved for sale in the European Union for the approximation of tissue layers where subcutaneous dead space exists between the tissue planes in large flap surgical procedures such as abdominoplasty and is being utilized in Europe to eliminate drains or reduce complications in patients undergoing large flap surgical procedures such as abdominoplasty, mastectomy, ventral hernia repair, decubitus and latissimus dorsi flap procedures. The company’s second product under development, Sylys Surgical Sealant, the first synthetic sealant designed specifically to help reduce anastomotic leaks, has received CE Mark approval in Europe as an adjunct to standard closure in ileostomy reversal procedures. TissuGlu and Sylys are the first products in a pipeline of technology that includes adhesives for surgical mesh fixation, meniscal repair and other orthopedic indications. Sylys and the other Cohera Medical products are currently available for investigational use only and have not yet been approved for sale by the U.S. Food and Drug Administration.
The European study, with four centers in Germany and seven in the United Kingdom, started in February and has completed enrollment of nearly 80 patients, with half of the patients treated with post-surgical drains and the other half treated with TissuGlu and no drains. After a 90-day follow up period, the study will assess the number of clinical interventions related to wound healing and fluid management in both groups. Preliminary assessments indicate that the outcomes of the study are positive.
“Our patients have been very enthusiastic about participating in the study when they hear about the chance of a drain-free procedure,” said Polly King, principal investigator at the Royal Cornwall hospital in the United Kingdom, which recruited the highest number of patients for the study. “Patients without drains feel more comfortable about going home sooner than those who wake up from surgery with a drain.”
“TissuGlu has been used for flap fixation after mastectomy and several other ‘flap procedures’ in Europe for a few years now. The body of evidence required to bring this new approach into the mainstream has been steadily growing, and this multinational prospective randomized study really represents the ‘Gold Standard’ of clinical evidence,” said Mart Pearson, vice president of Europe for Cohera Medical Inc. “We hope that these results will open the door for many other breast cancer patients who would greatly prefer a recovery without drains.”
This study adds to the growing body of evidence of positive TissuGlu outcomes.
Cohera Medical Inc. is developing and commercializing a line of surgical adhesives and sealants. Cohera Medical’s products are based on a chemical design that is resorbable, non-toxic, and easy-to-use. The company’s lead product, TissuGlu Surgical Adhesive, is indicated for use in the United States for the approximation of tissue planes in abdominoplasty procedures. TissuGlu is currently approved for sale in the European Union for the approximation of tissue layers where subcutaneous dead space exists between the tissue planes in large flap surgical procedures such as abdominoplasty and is being utilized in Europe to eliminate drains or reduce complications in patients undergoing large flap surgical procedures such as abdominoplasty, mastectomy, ventral hernia repair, decubitus and latissimus dorsi flap procedures. The company’s second product under development, Sylys Surgical Sealant, the first synthetic sealant designed specifically to help reduce anastomotic leaks, has received CE Mark approval in Europe as an adjunct to standard closure in ileostomy reversal procedures. TissuGlu and Sylys are the first products in a pipeline of technology that includes adhesives for surgical mesh fixation, meniscal repair and other orthopedic indications. Sylys and the other Cohera Medical products are currently available for investigational use only and have not yet been approved for sale by the U.S. Food and Drug Administration.