Mauna Kea Technologies10.30.17
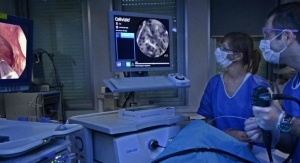
Mauna Kea Technologies, inventor of Cellvizio, the multidisciplinary confocal laser endomicroscopy platform, has received CE Mark for its CelioFlex UHD Confocal Miniprobes for use with Cellvizio in robotic-assisted surgery procedures. The company announced in August that it received U.S. Food and Drug Administration (FDA) 510(k) clearance for the same indication. The intended use of CelioFlex UHD Confocal Miniprobes is to provide visualization of body cavities, organs, and canals during endoscopic and laparoscopic surgical procedures, including robotic-assisted procedures.
Robotic-assisted surgery allows doctors to perform many types of complex procedures with greater precision, flexibility and control than is possible with conventional techniques. There are more than 700 Da Vinci robotic surgical systems installed in Europe, with more than 100,000 robotic-assisted procedures performed on these systems in 2016.1
Sacha Loiseau, Ph.D., founder and CEO of Mauna Kea Technologies, added, “This new CE Mark is a key regulatory and product milestone for the company, building on years of development and efforts to adapt its unique endomicroscopy platform to a wide variety of clinical settings. This includes surgical robotics, which is the fastest growing segment in surgery. This demonstrates our ability to strengthening Cellvizio’s value proposition and to expanding its addressable market opportunity, such as with our worldwide commercial partner in urology, Cook Medical. Following the recent FDA clearance for the same robotic-assisted procedure indication in the U.S, we have received positive feedback from and we look forward to advancing Cellvizio in this application in Europe.”
Mauna Kea Technologies is a global medical device company focused on eliminating uncertainties related to the diagnosis and treatment of cancer and other diseases thanks to real time in vivo microscopic visualization. The company’s flagship product, Cellvizio, has received clearance to sell a wide range of applications in more than 40 countries, including the United States, Europe, Japan, China, Canada, Brazil and Mexico. Mauna Kea is headquartered in Paris, France.
Reference
1. Intuitive Surgical Q3 2017 investor presentation.
Robotic-assisted surgery allows doctors to perform many types of complex procedures with greater precision, flexibility and control than is possible with conventional techniques. There are more than 700 Da Vinci robotic surgical systems installed in Europe, with more than 100,000 robotic-assisted procedures performed on these systems in 2016.1
Sacha Loiseau, Ph.D., founder and CEO of Mauna Kea Technologies, added, “This new CE Mark is a key regulatory and product milestone for the company, building on years of development and efforts to adapt its unique endomicroscopy platform to a wide variety of clinical settings. This includes surgical robotics, which is the fastest growing segment in surgery. This demonstrates our ability to strengthening Cellvizio’s value proposition and to expanding its addressable market opportunity, such as with our worldwide commercial partner in urology, Cook Medical. Following the recent FDA clearance for the same robotic-assisted procedure indication in the U.S, we have received positive feedback from and we look forward to advancing Cellvizio in this application in Europe.”
Mauna Kea Technologies is a global medical device company focused on eliminating uncertainties related to the diagnosis and treatment of cancer and other diseases thanks to real time in vivo microscopic visualization. The company’s flagship product, Cellvizio, has received clearance to sell a wide range of applications in more than 40 countries, including the United States, Europe, Japan, China, Canada, Brazil and Mexico. Mauna Kea is headquartered in Paris, France.
Reference
1. Intuitive Surgical Q3 2017 investor presentation.