Business Wire10.11.17
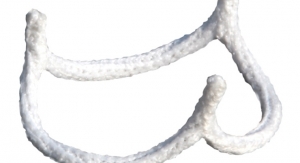
BioStable Science & Engineering Inc. has announced the first commercial use of the HAART 200 Aortic Annuloplasty Device in the United States by doctors at the West Virginia University (WVU) Heart and Vascular Institute in Morgantown, W.V. Drs. Lawrence Wei, Vinay Badhwar, and J. Scott Rankin were the collaborating surgeons for the procedure. The HAART 200 Aortic Annuloplasty Device is the only U.S. Food and Drug Administration (FDA)-cleared annuloplasty device designed to facilitate bicuspid aortic valve repair. The WVU Heart and Vascular Institute is the first of a select group of centers expected to participate in a limited release of the product this year.
Wei, director of the Center for Aortic Valve Disease and co-director of the Center for Aortic Surgery, was also the first U.S. surgeon to implant the HAART 300 Aortic Annuloplasty Device after that product received FDA clearance in March. Commenting on his experience, Wei said the HAART devices "remodel the valve annulus to facilitate leaflet reconstruction and optimize valve competence. These devices have expanded our ability to offer valve repair as an alternative to valve replacement for patients with aortic valve disease. Valve repair is the preferred treatment option for patients with appropriate indications and we are pleased to now have the ability to offer these solutions to our patients.”
“Surgeon interest in the HAART 200 Aortic Annuloplasty Device has been remarkably high because of the potential benefits of valve repair in this young patient population and the technical challenges of existing repair methods,” added Dr. Joe Cunningham, BioStable board member and managing director of lead investor, Santé Ventures. “FDA clearance of the HAART 200 Aortic Annuloplasty Device is a tremendous milestone that greatly expands the market opportunity for BioStable in the U.S.”
John Wheeler, President and CEO of BioStable Science & Engineering, concluded by noting, “Valve repair is widely recognized as the treatment of choice for patients with mitral valve disease. We believe the HAART Aortic Annuloplasty Devices will be the technologies that unlock the benefits of valve repair for patients with aortic valve disease and are very pleased to have our full product line available in the U.S.”
BioStable Science & Engineering is a cardiovascular device company developing and commercializing proprietary valve repair technologies that provide an alternative to valve replacement for patients with aortic valve disease. The company’s HAART Aortic Repair Technologies are designed to simplify and standardize aortic valve repair, enabling surgeons to offer the recognized clinical benefits of valve repair to patients undergoing surgical correction of aortic insufficiency or aortic root aneurysm.
The HAART 200 Aortic Annuloplasty Device is not available for sale outside of the United States.
Wei, director of the Center for Aortic Valve Disease and co-director of the Center for Aortic Surgery, was also the first U.S. surgeon to implant the HAART 300 Aortic Annuloplasty Device after that product received FDA clearance in March. Commenting on his experience, Wei said the HAART devices "remodel the valve annulus to facilitate leaflet reconstruction and optimize valve competence. These devices have expanded our ability to offer valve repair as an alternative to valve replacement for patients with aortic valve disease. Valve repair is the preferred treatment option for patients with appropriate indications and we are pleased to now have the ability to offer these solutions to our patients.”
“Surgeon interest in the HAART 200 Aortic Annuloplasty Device has been remarkably high because of the potential benefits of valve repair in this young patient population and the technical challenges of existing repair methods,” added Dr. Joe Cunningham, BioStable board member and managing director of lead investor, Santé Ventures. “FDA clearance of the HAART 200 Aortic Annuloplasty Device is a tremendous milestone that greatly expands the market opportunity for BioStable in the U.S.”
John Wheeler, President and CEO of BioStable Science & Engineering, concluded by noting, “Valve repair is widely recognized as the treatment of choice for patients with mitral valve disease. We believe the HAART Aortic Annuloplasty Devices will be the technologies that unlock the benefits of valve repair for patients with aortic valve disease and are very pleased to have our full product line available in the U.S.”
BioStable Science & Engineering is a cardiovascular device company developing and commercializing proprietary valve repair technologies that provide an alternative to valve replacement for patients with aortic valve disease. The company’s HAART Aortic Repair Technologies are designed to simplify and standardize aortic valve repair, enabling surgeons to offer the recognized clinical benefits of valve repair to patients undergoing surgical correction of aortic insufficiency or aortic root aneurysm.
The HAART 200 Aortic Annuloplasty Device is not available for sale outside of the United States.