Business Wire10.06.17
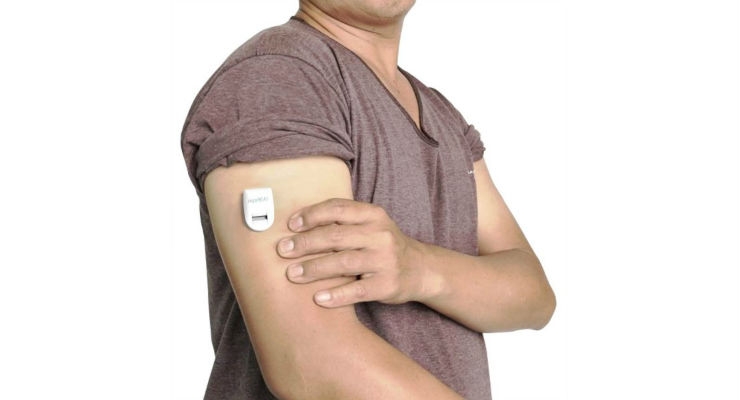
Nemaura Medical Inc., a medical technology company developing and commercializing sugarBEAT, a wireless adhesive skin patch for adjunctive use by diabetics as a non-invasive and needle-free glucose monitoring system, has announced interim clinical trial results for the sugarBEAT European clinical trial program.
This clinical trial program consists of a total of 525 patient days, comprising 75 patients continuously wearing sugarBEAT for up to 14 hours per day over seven consecutive days in a combination of home and clinic settings. The trials are expected to determine safety, accuracy, precision and utility of sugarBEAT as an adjunct to finger stick testing, and is expected to be completed over the next 10 weeks.
Interim results based on 175 patient days completed thus far have detected no skin irritation or serious adverse events. Minor adverse events were reported in the form of skin-patch de-lamination due to high humidity weather conditions, and were addressed by using an over-strip of adhesive or use of elastic arm band. Nemaura anticipates launching sugarBEAT in the United Kingdom shortly after completion of the clinical trial program.
The data generated thus far has indicated accuracy that is comparable to, and non-inferior to that previously achieved by the wired wrist worn predecessor SugarBEAT device, which received a CE mark in 2016.
SugarBEAT consists of a disposable skin-patch connected to a rechargeable transmitter, with a mobile app displaying glucose readings at regular five minute intervals.
Further clinical studies are planned including those for U.S. Food and Drug Administration approval, and to investigate the use of sugarBEAT in critical care settings.
Nemaura Medical is developing the wireless sugarBEAT disposable adhesive skin-patch as a non-invasive, needle-free, pain-free and affordable glucose monitoring system for adjunctive use by diabetics.
This clinical trial program consists of a total of 525 patient days, comprising 75 patients continuously wearing sugarBEAT for up to 14 hours per day over seven consecutive days in a combination of home and clinic settings. The trials are expected to determine safety, accuracy, precision and utility of sugarBEAT as an adjunct to finger stick testing, and is expected to be completed over the next 10 weeks.
Interim results based on 175 patient days completed thus far have detected no skin irritation or serious adverse events. Minor adverse events were reported in the form of skin-patch de-lamination due to high humidity weather conditions, and were addressed by using an over-strip of adhesive or use of elastic arm band. Nemaura anticipates launching sugarBEAT in the United Kingdom shortly after completion of the clinical trial program.
The data generated thus far has indicated accuracy that is comparable to, and non-inferior to that previously achieved by the wired wrist worn predecessor SugarBEAT device, which received a CE mark in 2016.
SugarBEAT consists of a disposable skin-patch connected to a rechargeable transmitter, with a mobile app displaying glucose readings at regular five minute intervals.
Further clinical studies are planned including those for U.S. Food and Drug Administration approval, and to investigate the use of sugarBEAT in critical care settings.
Nemaura Medical is developing the wireless sugarBEAT disposable adhesive skin-patch as a non-invasive, needle-free, pain-free and affordable glucose monitoring system for adjunctive use by diabetics.