I-MED Pharma10.04.17
I-MED Pharma has registered its i-Pen Tear Osmolarity System (i-Pen) in Japan. The i-Pen is an in-vivo handheld tear osmolarity device for the diagnosis and monitoring of dry eye disease. The system is currently approved for use in Canada and it holds CE Marking in the European Union (EU).
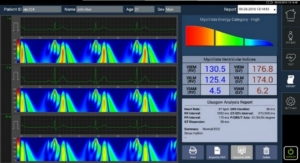
The i-Pen is a handheld platform that offers eye care professionals a quick, and reliable tool for screening dry eye patients. Measurements are taken by touching the device to the inside of the tear-soaked conjunctiva on the inside lower eyelid for two to four seconds. During that time, the i-Pen takes 192 readings and then displays the result on its LCD screen.
Daniel Hofmann, president of I-MED Pharma said, “The i-Pen device helps eye care professionals quickly and affordably assess a patient’s tear osmolarity, one of the important steps in identifying and quantifying the presence of dry eye disease. We would like to congratulate our Japanese partner, MORIA Japan K.K., for their successful registration of the i-Pen. Japan now joins our rapidly growing network of distribution partners with the system available in over a dozen countries worldwide.”
I-MED Pharma also announced that its second-generation device with an additional two biomarkers is entering the final phase of clinical testing. Initial applications for regulatory approval planned for the second half of 2018.
I-MED Pharma is a privately held Canadian company headquartered in Montreal, Quebec, servicing Canadian ophthalmologists, optometrists and the global eye care community. Established over 25 years ago, I-MED Pharma creates and distributes medical, surgical, and veterinary eye care products. The company researches, develops, and sources advanced solutions to eye disorders like cataracts, corneal degenerations, dry eye, glaucoma and Meibomian gland disease. Most recently, I-MED Pharma launched the world’s first hand-held in-vivo tear osmolarity test, the i-Pen osmolarity device. I-MED’s ocular surface disease product range includes diagnostic tools, ocular hygiene, nutrition, dry eye drops and ocular occlusion devices.
The i-Pen is a handheld platform that offers eye care professionals a quick, and reliable tool for screening dry eye patients. Measurements are taken by touching the device to the inside of the tear-soaked conjunctiva on the inside lower eyelid for two to four seconds. During that time, the i-Pen takes 192 readings and then displays the result on its LCD screen.
Daniel Hofmann, president of I-MED Pharma said, “The i-Pen device helps eye care professionals quickly and affordably assess a patient’s tear osmolarity, one of the important steps in identifying and quantifying the presence of dry eye disease. We would like to congratulate our Japanese partner, MORIA Japan K.K., for their successful registration of the i-Pen. Japan now joins our rapidly growing network of distribution partners with the system available in over a dozen countries worldwide.”
I-MED Pharma also announced that its second-generation device with an additional two biomarkers is entering the final phase of clinical testing. Initial applications for regulatory approval planned for the second half of 2018.
I-MED Pharma is a privately held Canadian company headquartered in Montreal, Quebec, servicing Canadian ophthalmologists, optometrists and the global eye care community. Established over 25 years ago, I-MED Pharma creates and distributes medical, surgical, and veterinary eye care products. The company researches, develops, and sources advanced solutions to eye disorders like cataracts, corneal degenerations, dry eye, glaucoma and Meibomian gland disease. Most recently, I-MED Pharma launched the world’s first hand-held in-vivo tear osmolarity test, the i-Pen osmolarity device. I-MED’s ocular surface disease product range includes diagnostic tools, ocular hygiene, nutrition, dry eye drops and ocular occlusion devices.