Business Wire08.14.17
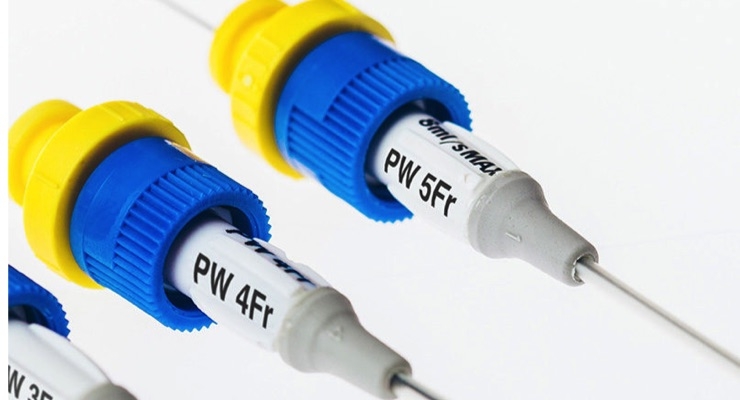
Access Scientific LLC has released a new best-in-class vascular access device—the POWERWAND REC (Rapid Exchange Catheter). This device, boasting Access's Zero-Edge Transitions on both the catheter and dilator, requires no skin nick enabling clinicians to exchange an existing small bore catheter for the large bore, high-flow (130ml/min) POWERWAND REC in a matter of seconds—saving the lives of patients in shock or impending shock.
"Central venous and intraosseous access are both useful tools in an emergency," noted Steve Bierman M.D., the company's chief medical officer and a former emergency physician. "But both of these procedures take significant time, entail substantial risk and result in flow rates far inferior to the POWERWAND Rapid Exchange Catheter."
When patients are brought into the emergency department or anesthesia in shock or impending shock, and they already have a small IV catheter in place, the POWERWAND REC is a more effective vascular access device, according to the company. The POWERWAND REC is quicker to insert, carries less risk, is power-injectable and delivers higher flow rates than a central venous and intraosseous access, executives said.
Decades ago, similar exchange catheters were introduced. However, these devices are made of Teflon-type material that crimps or kinks, and almost always require a surgical skin incision for proper placement—risking bleeding and infection. Moreover, the old devices are not power-injectable, often rendering them useless when emergency computed tomography scans are needed.
The POWERWAND REC is made of ChronoFlex C—the same material used for the POWERWAND midlines. This patented copolymer undergoes Access’s proprietary manufacturing processes, which render the ChronoFlex C kink-resistant, blood-drawable, high-flow and supported by over 20,000 catheter-days of peer-reviewed, published evidence with ZERO bloodstream infections.
"We will introduce two more state-of-the-art products to the POWERWAND family, for use by emergency physicians, anesthesiologists and rapid response teams in the near future," said Phil Royston, the company's president. "We already offer best-in-class power-injectable midlines and extended dwell catheters with the lowest complication and highest completion of therapy rates of any catheters in their class. Soon we will have a suite of best-in-class trauma products as well."
Access Scientific, based in San Diego, Calif., is an evidence-based, infection prevention company offering innovative vascular access devices designed to reduce the risk of vascular device-related complications, including bloodstream infections.
"Central venous and intraosseous access are both useful tools in an emergency," noted Steve Bierman M.D., the company's chief medical officer and a former emergency physician. "But both of these procedures take significant time, entail substantial risk and result in flow rates far inferior to the POWERWAND Rapid Exchange Catheter."
When patients are brought into the emergency department or anesthesia in shock or impending shock, and they already have a small IV catheter in place, the POWERWAND REC is a more effective vascular access device, according to the company. The POWERWAND REC is quicker to insert, carries less risk, is power-injectable and delivers higher flow rates than a central venous and intraosseous access, executives said.
Decades ago, similar exchange catheters were introduced. However, these devices are made of Teflon-type material that crimps or kinks, and almost always require a surgical skin incision for proper placement—risking bleeding and infection. Moreover, the old devices are not power-injectable, often rendering them useless when emergency computed tomography scans are needed.
The POWERWAND REC is made of ChronoFlex C—the same material used for the POWERWAND midlines. This patented copolymer undergoes Access’s proprietary manufacturing processes, which render the ChronoFlex C kink-resistant, blood-drawable, high-flow and supported by over 20,000 catheter-days of peer-reviewed, published evidence with ZERO bloodstream infections.
"We will introduce two more state-of-the-art products to the POWERWAND family, for use by emergency physicians, anesthesiologists and rapid response teams in the near future," said Phil Royston, the company's president. "We already offer best-in-class power-injectable midlines and extended dwell catheters with the lowest complication and highest completion of therapy rates of any catheters in their class. Soon we will have a suite of best-in-class trauma products as well."
Access Scientific, based in San Diego, Calif., is an evidence-based, infection prevention company offering innovative vascular access devices designed to reduce the risk of vascular device-related complications, including bloodstream infections.